DEFINITION
Plummer-Vinson is a syndrome of DYSPHAGIA with IRON-DEFICIENCY ANEMIA that is due to anomalies in the ESOPHAGUS (such as cervical esophageal webs).
It is known as Patterson-Kelly syndrome in the United Kingdom.
Definition 2
Definition 3
EPIDEMIOLOGY
FREQUENCY
Reliable prevalence data on Plummer-Vinson syndrome are lacking. In the first half of the 20th century, it was a relatively common finding, particularly among middle-aged Scandinavian women.
The rapid fall in prevalence of the syndrome in the latter part of the 20th century has paralleled an improvement in nutritional status, including widespread addition of iron to flour. The syndrome is now a rarity.
Webs may be found in 5-15% of selected patients presenting with dysphagia, but most of these patients do not have PVS.
RACE
Plummer-Vinson syndrome has mainly been described in whites.
SEX
Plummer-Vinson syndrome is more frequently observed in women. In earlier studies from Scandinavia, up to 90% of patients were women.
AGE
The typical age range at diagnosis is 40-70 years.
A handful of cases have been reported in children.
SYMPTOMS
Plummer-Vinson syndrome is rarely seen as a full-fledged syndrome.
Forme fruste occurrences are frequent. Virtually every manifestation of the syndrome is also seen as an isolated problem.
The postcricoid web, once considered pathognomonic of the syndrome, was shown epidemiologically to occur in only 15% of females with dysphagia.
The classical components of the syndrome include:
- SIDEROPENIA & HYPOCROMIC MICROCYTIC ANEMIA
Symptoms resulting from anemia (weakness, pallor, fatigue, tachycardia) may dominate the clinical
picture.

- DYSPHAGIA & ODINOPHAGIA
The dysphagia is usually painless and intermittent or progressive over years, limited to solids and
sometimes associated with weight loss.
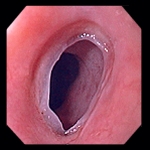
- POST-CRICOID WEBS
Their histological appearance is essentially that of folds of normal esophageal epithelium with some
underlying loose connective tissue.

- GASTRO-ESOPHAGEAL REFLUX & ESOPHAGITIS
- GASTRITIS & ACHLORHYDRIA
Anemia per se causes a superficial gastritis that, if untreated, progresses to atrophy of the gastric
mucosa.
Atrophy of the gastric mucosa causes achlorhydria.
- ATROPHIC GLOSSITIS
Plummer-Vinson syndrome sufferers often complain of a burning sensation with the tongue, and
atrophy of lingual papillae produces a smooth, shiny red tongue dorsum.

- CHEILITIS
Rhagades, angular stomatitis

- THEETH DAMAGE
Probably due to esophageal reflux, which erodes the enamel promoting caries formation
- SPLENOMEGALY
There is a 10% incidence of splenomegaly associated with sideropenic anemia.
The enlargement is minimal and pathogenesis is said to be unknown.
It seems likely that the reticuloendothelial system of the spleen merely exercises its normal function
and filters abnormal microcytic RBCs from the bloodstream.
- ENLARGED NODULAR THYROID GLAND
- KOILONYCHIA
Spoon-shaped finger nails.
It is not quite as clear that the koilonychia is due to anemia rather than low iron levels in the blood
serum.
Like the anemia, it clears when massive doses of iron are administered.
Nail changes are common in conditions with low O2 saturation levels – see clubbing in cyanotic
heart disease and chronic pulmonary disease – suggesting that anemia per se is the cause of the
nail disorder.

DIAGNOSIS
The diagnosis is based on the evidence of iron-deficiency anemia and one or more esophageal webs in a patient with postcricoid dysphagia.
HYSTOPATHOLOGY
The esophageal webs appear smooth, thin, and gray with eccentric or central lumen. They are characterized by one or more thin horizontal membranes consisting of squamous epithelium and submucosa.
Sometimes, chronic inflammatory cells may be observed in the submucosa. The webs typically occur in the proximal part of the esophagus. They usually protrude from the anterior wall, extending laterally but not to the posterior wall, which means that they rarely encircle the lumen.
LABORATORY TESTS
Laboratory examinations typically reveal iron deficiency anemia (hypochromic microcytic) with decreased values of hemoglobin, hematocrit, mean corpuscular volume, serum iron and ferritin, and increased total iron binding capacity.
RADIOLOGY
Esophageal webs can be detected by:
- barium swallow X-ray
- videofluoroscopy
- upper gastrointestinal endoscopy
DIFFERENTIAL DIAGNOSIS
Since dysphagia is a main clinical feature of Plummer-Vinson syndrome, the differential diagnosis includes all other causes of dysphagia especially malignant tumors, benign strictures or esophageal rings. Other reasons for dysphagia are diverticula, motility disorders such as achalasia, spastic motility disorders, scleroderma, diabetes mellitus, gastroesophageal reflux disease, and neuromuscular and skeletal muscle disorders.
PATHOGENESIS
Several etiopathogenic mechanisms have been postulated:
1) IRON DEFICIENCY
This is the prevalent theory.
The depletion of iron-dependent oxidative enzymes may produce myasthenic changes in muscles
involved in the swallowing mechanism, atrophy of the esophageal mucosa, and formation of webs as
epithelial complications.
The improvement in dysphagia after iron therapy provides evidence for an association between iron
deficiency and postcricoid dysphagia.
Moreover, the decline in Plummer-Vinson syndrome seems to parallel an improvement in nutritional
status, including iron supplementation.
However, the iron deficiency theory does not explain the predilection of webs for the upper
esophagus and the rarity of the syndrome in populations in which chronic iron deficiency is endemic
(eg, Africa).
2) AUTOIMMUNE PHENOMENON
The syndrome has been associated with autoimmune conditions, such as rheumatoid arthritis,
pernicious anemia, celiac disease, and thyroiditis.
3) GENETIC PREDISPOSITION
4) MALNUTRITION
Maybe vitamin B complex deficiency is involved as well.
FOCUS ON BIOCHEMICAL ALTERATIONS IN PLUMMER-VINSON SYNDROME:
Iron has a key-role in the respiratory chain, as the basic structure of cytochromes is the eme group.
The respiratory chain takes place on the mitochondrial membrane, involving 4 complexes.
Complex I (NADH dehydrogenase) removes two electrons from NADH and transfers them to a lipid-soluble carrier, ubiquinone (coenzyme Q). At the same time, Complex I moves protons (H+) across the membrane, producing a proton gradient.
Complex II (succinate dehydrogenase) is not a proton pump. It removes electrons from succinate and transfers them (via FAD) to coenzyme Q.
Complex III (cytochrome bc1 complex) removes two electrons from coenzyme Q and sequentially transfers them to two molecules of cytochrome c. At the same time, it moves protons across the membrane, producing a proton gradient.
Complex IV (cytochrome c oxidase) removes four electrons from four molecules of cytochrome c and transfers them to molecular oxygen (O2), producing two molecules of water (H2O). At the same time, it moves four protons across the membrane, producing a proton gradient.
As a result of all these steps, there is a proton-gradient across the mitochondrial membrane.
This gradient is used to move a specific H+-ATPase, a membrane pump that pumps protons out of mithocondrium and meanwhile builds up ATP molecules.
In absence of iron, no eme groups can be synthetized; this means there are no cytocromes and the respiratory chain cannot work.
The result is ATP depletion.
ATP is absolutely needful for cell survival.
For example, one critical spot is Calcium-ATPase:
calcium concentration must be kept quite low in cytosol.
In fact cells contain many phosphate ions, which would combine with calcium building up unsoluble complexes.
To avoid this, cells actively pump calcium out through a calcium-ATPase (this means with consumption of energy).
In case of ATP deficiency, calcium accumulates in cytosol and brings to cell death.
EXPLANATION OF THE MAIN SYMPTOMPS:
• ANEMIA:
iron deficiency brings to eme deficiency; as a result, there is hemoglobyn deficiency.
Red blood cells contain a small amount of hemoglobyn and appear mycrocitic and hypocromic.
• ESOPHAGEAL WEBS:
Calcium-ATPase is tipically present on sarcoplasmatic reticulum of muscle cells.
ATP is used to pump calcium in the sarcoplasmatic reticulum; then it is used for muscle contraction.
In case of ATP deficiency secondary to iron deficiency, muscle activity in esophagus is compromized.
Formation of webs might be an epithelial complication.
• ACHLORHYDRIA:
Gastric secretion is stimulated by 3 different agonists: acetylcholine, gastrin and histamine.
They all lead to activation of H/K-ATPase on apical membrane: H+ is actively pumped out of gastric cells, while Cl- follows passively to create HCl.
In case of ATP deficiency, there is also a reduction in HCl secretion.

PATIENT RISK FACTORS
GENETIC
Predisposition
ACQUIRED
Iron deficiency
HORMONAL
Women are at higher risk than men.
Menstrual blood loss (as much as 200-500 cc per month) might explain the sex ratio (higher rate of
sideropenic anemia among women), but Plummer-Vinson syndrome is more likley to occour after the
age of 40 than in fertile women.
So there might be also a different role of female hormones.
TISSUE SPECIFIC RISK FACTORS
ANATOMICAL
The alimentary tract is susceptible to iron deficiency; it rapidly loses iron-dependent enzymes due to its
high cell turnover, which is speculated to cause mucosa degeneration and web formation.
COMPLICATIONS
The Plummer-Vinson syndrome is one of the risk factors for developing squamous cell carcinoma of upper esophagus.