Definition:
Collagen is a group of naturally occurring proteins. In nature, it is found exclusively in animals. It is the main protein of connective tissue and skin. It is the most abundant protein in mammals,making up about 25% to 35% of the whole-body protein content.
There are at least 30 different collagen genes dispersed through the human genome. These 30 genes generate proteins that combine in a variety of ways to create over 20 different types of collagen fibrils. Types I, II and III are the most abundant and form fibrils of similar structure. Type IV collagen forms a two-dimensional reticulum and is a major component of the basal lamina. Collagens are predominantly synthesized by fibroblasts but epithelial cells also synthesize these proteins.
Molecular structure and images:
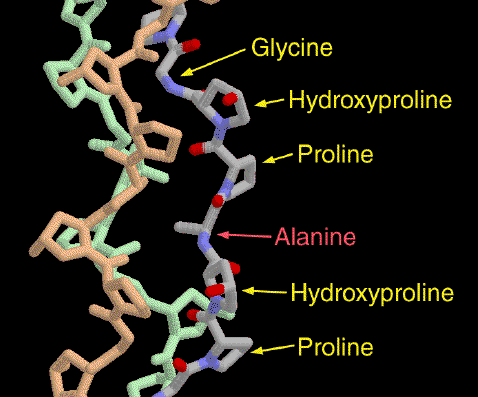
Collagen is a rigid structure, due to the Proline and Hydroxiproline presence.
The "tropocollagen" is collagen's structural unit and its molecular weight is about 285 kDa.
It is approximately 300 nm long and 1.5 nm in diameter, made up of three polypeptide strands (called alpha chains), each possessing the conformation of a left-handed helix.
Wikipedia
Structure
Percentage of aminoacids:
- Glycine (Gly) is found at almost every third residue
- Proline (Pro) makes up about 9% of collagen
- Hydroxyproline (Hyp), derived from proline.
- Hydroxylysine, derived from lysine (Depending on the type of collagen,varying numbers of
hydroxylysines have disaccharides attached to them)
Genes:
Synthesis and turnover:
Synthesis:

Most collagen forms in a similar manner, but the following process is typical for type I:
1. Inside the cell:
- Three peptide chains are formed (2 alpha-1 and 1 alpha-2 chain) in ribosomes along the rough endoplasmic reticulum (RER).
- These peptide chains (known as preprocollagen) have registration peptides on each end; and a signal peptide is also attached to each
- Peptide chains are sent into the lumen of the RER
- Signal Peptides are cleaved inside the RER and the chains are now known as procollagen
- Hydroxylation of lysine and proline amino acids occurs inside the lumen. This process is dependent on ascorbic acid (Vitamin C) as a cofactor
- Glycosylation of specific hydroxylated amino acid occurs
- Triple helical structure is formed inside the Endoplasmic Reticulum
- Procollagen is shipped to the Golgi apparatus, where it is packaged and secreted by exocytosis
2. Outside the cell:
- Registration peptides are cleaved and tropocollagen is formed by procollagen peptidase.
- Multiple tropocollagen molecules form collagen fibrils, via covalent cross-linking by lysyl oxidase which links hydroxylysine and lysine residues.
- Multiple collagen fibrils form into collagen fibers
- Collagen is attached to cell membranes via several types of protein, including fibronectin and integrin.
More detailed synthesis and synthesis regulation
Degradation and turnover
Types of collagene:


Localization and function:
Collagen is a primary component of the connective tissue located in the dermis, the tough inner layer of the skin. This kind of connective tissue is also found in mucous membranes, nerves, blood vessels, and organs. Collagen in these structures imparts strength, support, and a certain amount of elasticity.
Collagen is also a component of a kind of connective tissue that surrounds organs. This connective tissue encases and protects delicate organs like the kidneys and spleen.
Other locations where collagen fibers are prominent are in the tendons and ligaments.
Collagen adds strength to tendons and ligaments, and it imparts some stretch to these structures by allowing for some flexibility.
Cartilage is a connective tissue found in various places throughout the body, including the tip of the nose, the outside of the ears, the knees, and parts of the larynx and trachea. Cartilage consists of collagen fibers and cartilage cells. At these locations, collagen provides flexibility, support, and movement.

Main related disorders:
Medical use:
Collagens are widely employed in the construction of artificial skin substitutes used in the management of severe burns. These collagens may be derived from bovine, equine or porcine, and even human, sources and are sometimes used in combination with silicones, glycosaminoglycans, fibroblasts, growth factors and other substances.
Collagen is also sold commercially as a joint mobility supplement.
Collagen implant materials have a wide spectrum of clinical applications in medicine and dentistry, especially for bleeding control after surgical procedures, but also in order to increase tissue quality during wound healing. Although collagen has antigenic properties as an implant material, its good biocompatibility and easy application in daily clinical practice make it important for different surgical procedures.
Collagen is used as collagen membranes and collagenous sponge material and has the following clinical properties:
- Promote fibrobast chemotaxis
- Facilitate early connective tissue and epidermal wound healing
- Support periodontal regeneration
- Is hemostatic and possesses as good adherence to any wound bed
- Covering the whole wound area,protects it against infectious agens and against the loss of water and tissue fluids
- Covers the wound area, reducing or eliminating pain
- Promotes epithelial cells proliferation, development of basal membrane structures, a normal epidermal differentiation and enhancecs denaturation of epidermis which covers the healing wound
Collagen in dentistry:
- The periodontal ligament, commonly abbreviated as the PDL , is a group of specialized connective tissue fibers that essentially attach a tooth to the alveolar bone within which it sits.
In addition to the PDL fibers (I,J,K) , there is another set of fibers, known as the gingival fibers (H), which attach the teeth to their adjacent gingival tissue. Both the gingival fibers, as well as the PDL fibers, are composed primarily of type I collagen, although type III fibers are also involved.

- The PDL fibers (I,J,K) that insert into the cementum are called Sharpey's fibres .These fibres (bone fibres, or perforating fibres) are a matrix of connective tissue consisting of bundles of strong collagenous fibres connecting periosteum to bone. They are part of the outer fibrous layer of periosteum, entering into the outer circumferential and interstitial lamellae of bone tissue.

- Recent studies demonstrated that Type XVII collagen is a key player in tooth enamel formation.
Inherited tooth enamel hypoplasia occurs due to mutations in genes that encode major enamel components. Enamel hypoplasia also has been reported in junctional epidermolysis bullosa, caused by mutations in the genes that encode type XVII collagen (COL17), a component of the epithelial-mesenchymal junction. To elucidate the pathological mechanisms of the enamel hypoplasia that arise from the deficiency of epithelial-mesenchymal junction molecules, such as COL17, we investigated tooth formation in our recently established Col17 and Col17 rescued mice.
COL17 regulates ameloblast differentiation and is essential for normal formation of Tomes' processes. In conclusion, COL17 deficiency disrupts the epithelial-mesenchymal interactions, leading to both defective ameloblast differentiation and enamel malformation.
- Aminoacids in collagen I and III:
Type I
 p=.
p=. 
p=. 
Type III

Fore more details about the sequencing of Type III collagen view the complete study
Interaction of Smads with collagen types I, III, and V:
Ontogenesis of the mammalian orofacial region is controlled by numerous developmental signals, including those initiated by the transforming growth factors beta (TGFbetas). Targeted deletion of the genes encoding several of the TGFbetas in mice has been shown to result in clefts of the secondary palate. Members of the TGFbeta family of growth factors utilize intracellular Smads as signal transducers. Smads 2 and 3 are transcriptional regulators that bind DNA through their conserved MH1 domains and activate/inhibit transcription of TGFbeta-responsive genes through their MH2 domains. Using a least two-hybrid screen of a cDNA expression library constructed from fetal murine orofacial tissue, we have identified three types of collagens (types I, III, and V) that are capable of binding to the MH2 domain of Smad 3.Each collagen also bound to the MH2 domains of Smads 4 and 7 and, to a lesser extent, full-length Smads 1, 2, 3, and 4. Binding of Smads to collagen is a novel observation. Moreover, TGFbeta is a potent regulator of collagen synthesis and turnover during mammalian orofacial development. These data thus suggest an important means of feedback regulation of the TGFbeta signaling cascade.
Link to the original article
For more info on Smad-dependent stimulation of type I collagen gene