Fever (also known as pyrexia or controlled hyperthermia) is a common symptom characterized by an elevation of temperature above the normal range of 36.5–37.5 °C (98–100 °F)
Temperature circadian rythm
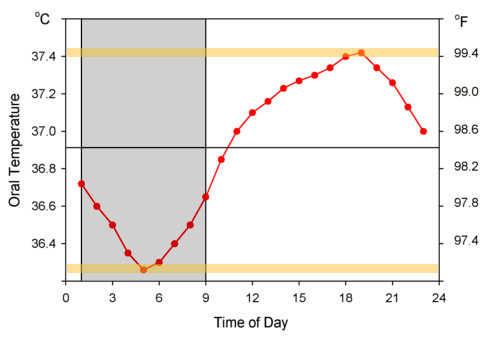
from

from
Pyrogenic stimuli
Fever may be provoked by many stimuli.
- exogenic pyrogens
- bacteria and their endotoxins, viruses, yeasts, spirochets, protozoa,
- immune reactions,
- several hormones,
- medications, and
- synthetic polynucleotides.
- endogenic pyrogens. Cells stimulated by exogenic pyrogens form and produce cytokines called endogenic pyrogens that centrally affect the thermosensitive neurons in the preoptic area of the hypothalamus increase the production of heat and decrease in heat loss. The body temperature increses until it reaches the set point. This information is transferred by temperature of blood that flows around the hypothalamus. The decrease of temperature is controlled by activation of mechanisms regulating increased outcome of heat to the surrounding area. Increased outcome continues in favourable case until the new equilibrium is achieved.
- IL-1,
- IL-6 and
- cachectin also called the tumour necrosis factor- (TNF-).
- interferons
These are glycoproteins that also have other important effects. They are produced especially by monocytes and macrophages but also by endothelial cells and astrocytes.
After administration an endotoxin in an experiment, the level of plasmatic TNF- increases and fever occurs. Increased concentrations of IL-1 and TNF- are also found in sepsis. The production of these cytokines is regulated by the positive feedback mechanism. Besides this, macrophages activated by IFN- may increase the production of IL-1 and TNF- primary induced by other stimuli. On the other hand, glucocorticoids and prostaglandins of group E may display inhibitory effect on the production of IL-1 and TNF- . Released IL-1 and TNF- are transported by blood. They affect the target cells in the close proximity or in distant sites. The target cells have specific receptors for IL-1 and TNF- . In the hypothalamus, IL-1 and TNF- trigger the synthesis of prostaglandis of group E from the arachidonic acid of cytoplasmic membranes of target cells. Precise mechanism by which prostaglandin PGE reset the central thermostat, is not known. Aspirin and the non-steroidal antiphlogistics display antipyretic activity by inhibiting the cyclo-oxygenase, an enzyme responsible for the synthesis of PGE (these antipyretics don't inhibit the production of TNF- or IL-1). Glucocorticoids work antipyretically by inhibiting the production of IL-1 and TNF- , and by inhibiting the metabolic processes of arachidonic acid.
Heat production in fever
Heat production is a side effect of oxidative metabolism in mitochondria
The main task in heat production has thermogenesis caused by the effect of thyroid hormones. Hormones of thyroid gland stimulate Na/K-ATPase found in cytoplasmic membranes. Increased production of heat is achieved by increasing the metabolic processes in which energy is released in the form of heat. The greatest importance is splitting of ATP when 54kJ are released from one mol of ATP.
Thyroid hormones and thermogenesis 1990

UCP and thermogenesis
Molecular understanding of adaptive thermogenesis 2000

Thermogenesis requires a `futile' cycle that depends on excess intracellular Ca++ or Na+ followed by activation of the corresponding pumps or their direct activation.
Stimulation by thyroid hormone of Na+-H+ exchange activity in cultured opossum kidney cells 1990
T3 is lower during infection 1977
The lowered T3 levels are not suggestive of a hypothyroid state, but perhaps could be due to decreased peripheral conversion of T4 to T3
Effects of hypertermia
HSP
Heat shock proteins are a class of functionally related proteins whose expression is increased when cells are exposed to elevated temperatures or other stress
Heat-shock proteins are named according to their molecular weight. For example, Hsp60, Hsp70 and Hsp90 (the most widely-studied HSPs) refer to families of heat shock proteins on the order of 60, 70 and 90 kilodaltons in size, respectively.
The small 8 kilodalton protein ubiquitin, which marks proteins for degradation, also has features of a heat shock protein.
