DEFINITION
Guillain-Barrè syndrome (also known as acute inflammatory demyelinating polyneuropathy, acute idiopathic polyradiculoneuritis, acute idiopathic polyneuritis, French Polio, Landry's ascending paralysis and Landry Guillain Barrè syndrome) is an acute, autoimmune inflammatory process affecting the peripheral nervous system and nerve roots. It results in demyelination.

The process is often preceded by a viral or bacterial infection, surgery, immunization, lymphoma, or exposure to toxins. However, 60% of cases do not have a known cause. Common clinical manifestations include progressive weakness, loss of sensation, and loss of deep tendon reflexes. Weakness of respiratory muscles and autonomic dysfunction may occur.
EPIDEMIOLOGY
Frequency
United States
Acute inflammatory demyelinating polyneuropathy is the most common acquired demyelinating polyneuropathy. The incidence is 0.6-1.7 cases per 100,000 per year. No significant seasonal variation has been noted.
International
Frequency is not well documented. Of 2 predominant Guillain-Barrè syndrome (GBS) subtypes, a demyelinating subtype (AIDP) predominates in the United States and Europe, and axonal subtype (AMAN) is the predominant form in China. Previous clinical studies suggested that AMAN also occurs in Mexican children. Similar outbreaks have been reported in Mexico, Spain, and Jordan.
Mortality/Morbidity
In 3 recent large studies, mortality rate ranged from 2-6%. In general, death is due to complications of ventilation. More than 75% of patients have complete or near-complete recovery with no deficit or only mild residual fatigue and distal weakness. Other patients, almost all of whom required ventilation, report severe dysesthesias or moderately severe distal weakness as residual symptoms. About 15% of patients end up with significant neurological residuals.
Race
Acute inflammatory demyelinating polyneuropathy occurs in all races and in all regions of the world.
Sex
The male-to-female ratio is 1.1-1.7:1.
Age
Patients have ranged in age from 2 months to 95 years.
• In the United States, age distribution is apparently bimodal, with most patients presenting from 15-35 years or 50-75 years.
• In China (and other countries), frequent outbreaks in children aged 2-12 years have been reported.
SYMPTOMS
The disorder is characterized by weakness which affects the lower limbs first, and rapidly progresses in an ascending fashion, accompanied or not by dysesthesias (numbness or tingling). As the weakness progresses upward, usually over periods of hours to days, the arms and facial muscles also become affected. Frequently, the lower cranial nerves may be affected, leading to bulbar weakness, (oropharyngeal dysphagia, that is difficulty with swallowing, drooling, and/or maintaining an open airway) and respiratory difficulties. Most patients require hospitalization and about 30% require ventlatory assistance. Facial weakness is also commonly a feature, but eye movement abnormalities are not commonly seen in ascending GBS, but are a prominent feature in the Miller-Fisher variant (see below). Sensory loss, if present, usually takes the form of loss of proprioception (position sense) and areflexia (complete loss of deep tendon reflexes), an important feature of GBS. Loss of pain and temperature sensation is usually mild. In fact, pain is a common symptom in GBS, presenting as deep aching pain, usually in the weakened muscles, which patients compare to the pain from overexercising. These pains are self-limited and should be treated with standard analgesics. Bladder dysfunction may occur in severe cases but should be transient. If severe, spinal cord disorder should be suspected.
Fever should not be present, and if it is, another cause should be suspected.
In severe cases of GBS, loss of autonomic function is common, manifesting as wide fluctuations in blood pressure, orthostatic hypotension, and cardiac arrhythmias.
CLINICAL VARIANTS
Six different subtypes of Guillain-Barrè syndrome (GBS) exist:
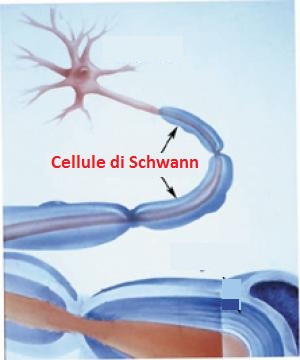
• Acute inflammatory demyelinating polyneuropathy (AIDP) is the most common form of GBS, and the term is often used synonymously with GBS. It is caused by an auto-immune response directed against Schwann cell membranes.
• Miller-Fisher Syndrome (MFS) is a rare variant of GBS and manifests as a descending paralysis, proceeding in the reverse order of the more common form of GBS. It usually affects the ocular muscles first and presents with the triad of ophthalmoplegia, ataxia, and areflexia. Anti-GQ1b antibodies are present in 90% of cases.
• Acute motor axonal neuropathy (AMAN), aka. Chinese Paralytic Syndrome, attacks motor nodes of Ranvier and is prevalent in China and Mexico. It is likely due to an auto-immune response directed against the axoplasm of peripheral nerves. The disease may be seasonal and recovery can be rapid. Anti-GD1a antibodies are present. Anti-GD3 antibodies are found more frequently in AMAN.
• Acute motor sensory axonal neuropathy (AMSAN) is similar to AMAN but also affects sensory nerves with severe axonal damage. Like AMAN, it is likely due to an auto-immune response directed against the axoplasm of peripheral nerves. Recovery is slow and often incomplete.
• Acute panautonomic neuropathy is the most rare variant of GBS, sometimes accompanied by encephalopathy. It is associated with a high mortality rate, due to cardiovascular involvement, and associated dysrhythmias. Impaired sweating, lack of tear formation, photophobia, dryness of nasal and oral mucosa, itching and peeling of skin, nausea, dysphagia, constipation unrelieved by laxatives or alternating with diarrhea occur frequently in this patient group. Initial nonspecific symptoms of lethargy, fatigue, headache, and decreased initiative are followed by autonomic symptoms including orthostatic lightheadedness, blurring of vision, abdominal pain, diarrhea, dryness of eyes, and disturbed micturition. The most common symptoms at onset are related to orthostatic intolerance, as well as gastrointestinal and sudomotor dysfunction. Parasympathetic impairment (abdominal pain, vomiting, obstipation, ileus, urinary retention, dilated unreactive pupils, loss of accommodation) may also be observed.
• Bickerstaff’s brainstem encephalitis (BBE), is further variant of Guillain-Barrè syndrome. It is characterized by acute onset of ophthalmoplegia, ataxia, disturbance of consciousness, hyperreflexia or Babinski’s sign. The course of the disease can be monophasic or remitting-relapsing. Large, irregular hyperintense lesions located mainly in the brainstem, especially in the pons, midbrain and medulla are described in the literature. BBE despite severe initial presentation usually has a good prognosis. MRI plays a critical role in the diagnosis of BBE.
DIAGNOSIS
The diagnosis of GBS usually depends on findings such as rapid development of muscle paralysis, areflexia, absence of fever, and a likely inciting event. CSF (cerebrospinal fluid) analysis and electrodiagnostics are used almost every time to verify symptoms, but because of the acute nature of the disorder, they may not become abnormal until after the first week of onset of signs and symptoms.
• CSF
Typical CSF findings include albumino-cytological dissociation. As opposed to infectious causes, this is an elevated protein level (100 - 1000 mg/dL), without an accompanying pleocytosis (increased cell count). A sustained pleocytosis may indicate an alternative diagnosis such as infection.
High protein level in CSF depends on a Blood-CSF Barrier dysfunction. This could be a consequence of a greater swelling in the region of the spinal roots, affecting CSF reabsorption through arachnoid villi into the veins present in this region with subsequent reduction of flow and accumulation of proteins and/or intense inflammation at the junction of the dorsal and ventral roots causing a breakdown in the blood-nerve (spinal root) barrier and the transudation of plasma proteins into the CSF. Signs of intrathecal synthesis of IgG have been reported in GBS patients with variable frequency, but it is mostly accepted that high CSF IgG concentration depends mainly on the degree of Blood-CSF Barrier dysfunction, thus excluding the intrathecal origin.
(An appraisal of blood-cerebrospinal fluid barrier dysfunction during the course of Guillain Barré syndrome).
• Electrodiagnostics
Electromyography (EMG) and nerve conduction study (NCS) may show prolonged distal latencies, conduction slowing, conduction block, and temporal dispersion of compound action potential in demyelinating cases. In primary axonal damage, the findings include reduced amplitude of the action potentials without conduction slowing.
• Biochemical screening: includes electrolyte levels; liver function tests (LFTs); CPK level; erythrocyte sedimentation rate (ESR); antiganglioside antibodies; and antibodies to C jejuni, cytomegalovirus, Ebstein-Barr virus, herpes simplex virus (HSV), HIV, and M pneumoniae.
• Stool culture for C. jejuni
• Imaging Studies
MRI
MRI is a sensitive but nonspecific test.
Selective anterior nerve root enhancement appears to be strongly suggestive of GBS.
The cauda equine nerve roots are enhanced in 83% of patients.
• Forced vital capacity
Forced vital capacity is very helpful in guiding disposition and therapy.
PATHOPHYSIOLOGY
All forms of Guillain-Barrè syndrome are due to an immune response to foreign antigens (such as infectious agents) which are mistargeted at host nerve tissues instead (a form of antigenic mimicry). The targets of such immune attack are thought to be gangliosides, which are complex glycosphingolipids present in large quantities on human nerve tissues, especially in the nodes of Ranvier. An example is the GM1 ganglioside, which can be affected in as many as 20-50% of cases, especially in those preceded by Campylobacter jejuni infections. Another example is the GQ1b ganglioside, which is the target in the Miller Fisher syndrome variant.
The most common antecedent infection is Campylobacter jejuni.

Molecular mimicry between C. jejuni lipooligosaccharides (LPS) and gangliosides plays a key role in the induction of antiganglioside antibodies and neurological symptomes in patients with GBS or MFS.
C. jejuni isolates from GBS and MFS patients more frequently express ganglioside mimics in their LPS than isolates from uncomplicated enteritis patients. Cross-reactive anti-LPS antiglycolipid responses can be detected in almost all culture proven cases of C. jejuni-related GBS and MFS but not in serum from patients with an uncomplicated enteritis. Differences in LPS structure determine the specificity of the antibody response and thereby the clinical features of the patients. (Structure of Campylobacter jejuni Lipopolysaccharides Determines Antiganglioside Specificity and Clinical Features of Guillain-Barré and Miller Fisher Patients)

Structure of C. jejuni LPS types and the corresponding ganglioside mimics. Symbols:
 N-acetylglucosamine,
N-acetylglucosamine,  heptose,
heptose,  3-deoxy-D-manno-octulosonic acid,
3-deoxy-D-manno-octulosonic acid,  2-aminoethyl phosphate.
2-aminoethyl phosphate.
An important feature in ganglioside mimicry is the presence of sialic acid (N-acetylneuraminic acid) in both LOS and gangliosides. Specific types of the C. jejuni LOS biosynthesis gene locus (“LOS locus”) are clearly associated with immune-mediated neuropathy (GBS) and with the presence of ganglioside-mimicking structures in the LOS. Genes involved in sialylation of the LOS are crucial for the induction of anti-ganglioside antibodies. (The crucial role of Campylobacter jejuni genes in anti-ganglioside antibody induction in Guillain-Barrè syndrome)
Enteric infection with Campylobacter results in an estimated 2 million cases of diarrhea per year in the United States. Sero-prevalence for anti-Campylobacter IgG antibodies increases with increasing age. The data from sero-surveillance studies indicate that both in developing and developed countries, frequent exposure to Campylobacter occurs, leading to a detectable antibody response. Antibody decay, although highly variable between individuals, was demonstrated in longitudinal studies. Hence, even though anti-Campylobacter IgG antibodies may persist for several years, their high prevalence indicates repeated (symptomless) infection by Campylobacter during life (Immunity to Campylobacter: its role in risk assessment and epidemiology).
Only a very small proportion of patients affected by C. jejuni infection develops GBS. This raises a series of question. Do LPSs from all or only some Campylobacter strains bear epitopes that cross-react with neural glycoconjugates? If all do, is there a quantitative difference between strains with respect to expression of these epitopes? Do all patients with Campylobacter infection develop antiganglioside antibodies, or is there a difference in the immune response of patients who develop GBS and those who do not?
All the C. jejuni isolates, both GBS- and non-GBS-associated, contained ganglioside-like epitopes known to be shared with nerve fibers. The sera from all of the GBS patients contained antiganglioside antibodies directed against one or more of these ganglioside-like epitopes. In contrast, the sera from the patients with diarrhea lacked only these antibodies. C. jejuni LPSs exhibit molecular mimicry with ganglioside-like moieties in peripheral nerve fibers, but they also indicate that infection with a Campylobacter organism expressing a neural cross-reactive epitope is not in itself sufficient to produce GBS. These differences in the immune responses could be due to differences in the hosts or in the organisms.
HOST RELATED FACTORS
A role for genetic susceptibility of the host is suggested by the observation that single individuals have been identified who had multiple episodes of acute GBS after enteritis and vaccination. The organisms associated with GBS could have a greater capacity to stimulate a humoral immune response than most Campylobacter organisms. The important point is that the immune response to LPS antigens is greater in the GBS group than in the diarrhea-only group. (Campylobacter jejuni lipopolysaccharides in Guillain-Barrè syndrome. Molecular mimicry and host susceptibility)
BACTERIUM RELATED FACTORS: ITS PATHOGENICITY RELATED TO IRON’S AVAILABILITY
The transcriptional response of C. jejuni to iron availability is pleiotropic, involving the differential expression of genes belonging to several functional groups, ranging from energy metabolism to cell surface structures to iron acquisition and to oxidative stress defense. Several of these genes encode proteins that have been shown to be involved in the mechanism of Campylobacter pathogenesis. The level of Campylobacter glycoproteins and/or the efficiency of glycan addition to those proteins was found to increase with iron limitation. A mutant deficient in the glycosylation process has been shown to be affected in its ability to colonize the mouse gastrointestinal tract. Consequently, iron availability likely modulates the abilities of Campylobacter to adhere to, invade, and colonize the host gastrointestinal tract. Thus, as in other pathogens, iron might constitute a key environmental signal for controlling Campylobacter pathogenesis. It’s necessary iron’s acquisition in the colonization process and survival of C. jejuni in vivo, as demonstrated by the existence of the CfrA protein (ferric enterobactin receptor). (Iron Acquisition and Regulation in Campylobacter jejuni)
The end result of autoimmune attack on the peripheral nerves is inflammation of myelin and conduction block, leading to a muscle paralysis that may be accompanied by sensory or autonomic disturbances. However, in mild cases, axonal function remains intact and recovery can be rapid if remyelination occurs. In severe cases, such as in the AMAN or AMSAN variants, axonal degeneration occurs, and recovery depends on axonal regeneration. Recovery becomes much slower, and there is a greater degree of residual damage.
POSSIBLE MECHANISMS OF NERVE CONDUCTION BLOCK:
- Autoantibodies to gangliosides such as GM1 have been proposed to disrupt nodal Nav channels (voltage-gated Na channels) and lead to Guillain-Barrè syndrome, especially to AMAN variant. At the acute phase with progressing limb weakness, Nav channel clusters were disrupted or disappeared at abnormally lengthened nodes concomitant with deposition of IgG and complement products. Nav channel alterations occur as a consequence of complement-mediated disruption of interactions between axons and Schwann cells. (Anti-GM1 Antibodies Cause Complement-Mediated Disruption of Sodium Channel Clusters in Peripheral Motor Nerve Fibers; The sera from GM1 ganglioside antibody positive patients with Guillain-Barré syndrome or chronic inflammatory demyelinating polyneuropathy blocks Na+ currents in rat single myelinated nerve fibers)

- Humoral factors (possibly cytokines), other than antibodies are secreted by lymphocytes and macrophages stimulated by gram negative LPS (e.g. Campylobacter jejuni-LPS), and cause functional conduction abnormalities when the blood–nerve barrier is disrupted. (Induction of conduction block by Campylobacter jejuni lipopolysaccharides and focal neural insult)
TREATMENT
Supportive care with monitoring of all vital functions is the cornerstone of successful management in the acute patient. Of greatest concern is respiratory failure due to paralysis of the diaphragm. Early intubation should be considered in any patient with a vital capacity <20 ml/kg. Once the patient is stabilized, treatment of the underlying condition should be initiated as soon as possible. Either high-dose intravenous immunoglobulins or plasmapheresis can be administered, as they are equally effective and a combination of the two is not significantly better than either alone. Therapy is no longer effective 2 weeks after the first motor; following the acute phase, the patient may also need rehabilitation to regain lost functions.