Description
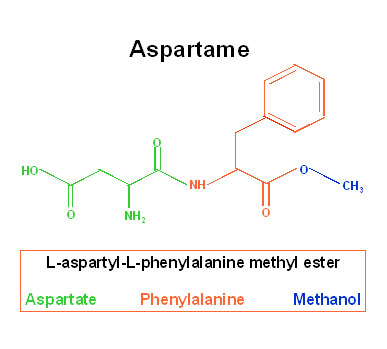
Aspartame, a synthetic nonnutritive sweetener, is a methyl ester of a dipeptide composed of aspartic acid and phenylalanin.
Aspartame is a white crystalline powder having no odor, but is intensely sweet. Aspartame is approximately 200 times sweeter than sucrose, the accepted standard for sweetness. Although aspartame yields the same caloric intake as sugar on a weight to weight basis (i.e., 4 kcal/g), it can be added at almost 200 times lower levels and achieve the same sweetness, thereby providing a far lower net caloric intake. This attribute has resulted in the use of aspartame as a low calorie or nonnutritive sweetener in foods and beverages worldwide.
There are two forms of aspartame, an alpha and a beta form, but only the α form is sweet.
Structure of aspartame
Metabolism
The intestinal absorption and metabolism of aspartame have been intensively studied, in rodents, pigs, primates and humans. In all species examined, aspartame is metabolized in the gastrointestinal tract by esterases and peptidases into three components: the two constituent amino acids, aspartic acid (40%) and phenylalanine (50%), and methanol (10%).
Aspartame may be completely hydrolyzed to these three components in the gastrointestinal lumen and absorbed into the general circulation, or may be hydrolyzed to methanol and aspartylphenylalanine dipeptide by intestinal esterases, possibly chymotrypsin. In this case, the dipeptide is absorbed into the gastrointestinal mucosa cells and then cleaved into amino acids.
Aspartame can also be absorbed into the mucosal cells prior to hydrolysis, and be cleaved within the cell, to its three components, which then enter circulation.
The available evidence indicates that aspartame does not enter the circulation prior to hydrolysis.
Phenylalanine enters the plasma free amino acid pool from the portal blood after partial conversion to tyrosine by hepatic phenylalanine hydroxylase.
Aspartate is metabolized within the enterocyte via transamination producing oxaloacetate, thereby reducing theconcentration of aspartate entering the portal circulation and plasma free amino acid pool.
Methanol is not subject to metabolism within the enterocyte and rapidly enters the portal circulation. The methanol is oxidized in the liver to formaldehyde. Enzymes involved depend on species: In the rat, the metabolism of methanol to formaldehyde is mediated though a catalase-peroxidase system, whereas in primates and humans, an alcohol dehydrogenase is responsible. Formaldehyde is further oxidized to formic acid by formaldehyde dehydrogenase. Formic acid is ultimately converted to CO2 and water, via the formation of 10-formyl tetrahydrofolate.
Metabolism of aspartame
Effect of aspartame on the brain
Berardi Daniela (matr. 300429) e Coppo Alessandra (matr. 276329 27)
Scuola di Specializzazione in Patologia Clinica 2° Anno