The aim of this topic is to try to explain the association between Deferoxamine iron-cheletion and sepsis sustained by Yersinia enterocolitica.

Yersinia enterocolitica is a species of Gram-negative coccobacillus-shaped bacterium, belonging to the family enterobacteriaceae, that most often causes enterocolitis, acute diarrhea and terminal ileitis, but, if it spreads systemically, can also results in fatal sepsis.
Yersinia genus includes also Y. Pestis and Y. Pseudotubercolosis.
Y. enterocolitica is a heterogeneous group of strains, which are classified into 6 biogroups, and into more than 57 O serogroups. The biotype 1B is the most virulent.
The major route of Y. enterocolitica infection is through contaminated foods or water, but it can also occur via blood transfusion.
PATHOGENESIS
Most isolates of Y. enterocolitica from food or clinical materials have either of two pathogenic properties
- First property is the ability to penetrate the intestinal wall, which is thought to be controlled by 70-kb virulence plasmid (pYV/pCD) genes; that is absent in avirulent strains
- The second one is the production of heat-stable enterotoxin which is controlled by chromosomal genes (ystA, ystB, and ystC).
Invasion of human epithelial cells and penetration of the mucosa occurs in the ileum, followed by multiplication in Peyer patches. A 103-kd protein, known as invasin and determined by the INV gene, mediates bacterial invasion. After invasion process, Yersinia defend the attack by resident macrophages by expressing an antiphagocytic strategy mediated by a plasmid encoded type III secretion, of three protein effectors, YopH, T, and E, that disrupt cytoskeletal assembly required for phagocytosis process .
Yersinia enterocolitica : Mode of Transmission, Molecular Insights of Virulence, and Pathogenesis of Infection
VIRULENCE FACTORS
- pYV
- HPI. High-Pathogenicity Island. (Only in higly pathogenic bioype 1B)
HPI encodes proteins that are involved in the biosynthesis, regulation, and transport of the siderophore yersiniabactin and has thus been referred to as an “iron capture island”. There are five main genes within this island (psn, irp1, irp2, ybtP, and ybtQ) that are involved in the yersiniabactin system [80, 82, 83]. This system is positively regulated by YtbA, which is, itself, negatively regulated by the iron-responsive regulator Fur . The psn and irp2 genes are important for the high-pathogenicity phenotype of YPT.
Pathogenesis of Y. enterocolitica and Y. pseudotuberculosis in Human Yersiniosis
YERSINIABACTIN & DEFEROXAMINE: TWO SIDEROPHORES
Siderophores, compounds of low molecular mass with high affinities for ferric iron, are important virulence factors in pathogenic bacteria. Iron, an essential element for life utilized by such cellular processes as respiration and DNA replication, is extensively chelated by host proteins like lactoferrin and ferritin; thus, the pathogen produces molecules with an even higher affinity for Fe3+ than these proteins in order to acquire sufficient iron for growth.
Yersiniabactin (Ybt)

is a siderophore found in the pathogenic bacteria Yersinia pestis, Yersinia pseudotuberculosis, and Yersinia enterocolitica, as well as several strains of enteropathogenic Escherichia coli. It is a four ring structure composed of carbon, hydrogen, nitrogen, oxygen, and sulfur. According to X-ray crystallography, it binds Fe3+as a 1:1 complex by three nitrogen electron pairs and three negatively charged oxygen atoms with a distorted octahedral structure.
The complex yersiniabactin-Fe3+ recognizes the specific bacterial outer membrane receptor TonB and is translocated with the help of membrane-embedded proteins into the cytosol where the iron is discharged from yersiniabactin and utilized in various metabolic pathways.
The availability of iron provides the bacteria with the ability to multiply in the host and to cause systemic infections.
Wikipedia-Yersiniabactin
Deferoxamine
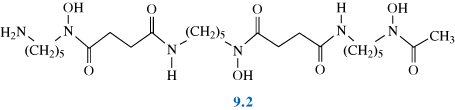
(also known as desferrioxamine B, desferoxamine B, DFO-B, DFOA, DFB or Desferal) is a bacterial siderophore produced by the actinobacteria Streptomyces pilosus. It has medical applications as a chelating agent used to remove excess iron from the body.
Deferoxamine chelates iron by forming a stable complex that prevents the iron from entering into further chemical reactions. It readily chelates iron from ferritin and hemosiderin but not readily from transferrin; it does not combine with the iron from cytochromes and hemoglobin. Theoretically, 100 parts by weight of Desferal is capable of binding approximately 8.5 parts by weight of ferric iron (molar ratio 1:1).
Wikipedia-Deferoxamine
Novartis Desferal
CONCLUSIONS:
In patient with iron overload and under therapy with Deferoxamine, Yersinia Enterocolitica infections can result in sepsis.
These patients have a greater availability of iron in the blood stream and in tissues to be used by the pathogens.
It is known that Y. Enterocolitica, can utilize siderophores produced by other bacteria, or somministrated as drugs, in particular Deferoxamine.
Medscape-Y.enterocolitica
We can assume that the similarity between Deferoxamine and Yersiniabactin could lead to an important increase of the YE virulence, bringing to a spread and more severe infection, and thus sepsis.