Papers
DNA methylation requires a DNMT1 ubiquitin interacting motif (UIM) and histone ubiquitination. 2015
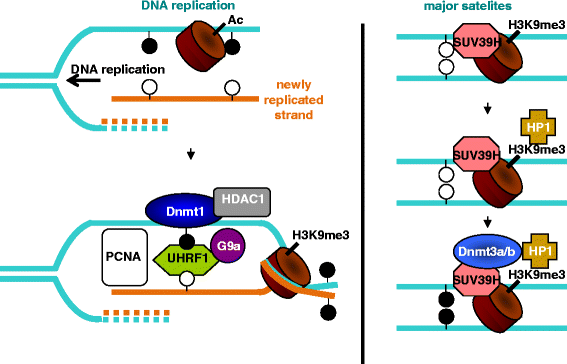
- DNMT1 is recruited by PCNA and UHRF1 to maintain DNA methylation after replication. UHRF1 recognizes hemimethylated DNA substrates via the SRA domain, but also repressive H3K9me3 histone marks with its TTD. With systematic mutagenesis and functional assays, we could show that chromatin binding further involved UHRF1 PHD binding to unmodified H3R2. These complementation assays clearly demonstrated that the ubiquitin ligase activity of the UHRF1 RING domain is required for maintenance DNA methylation. Mass spectrometry of UHRF1-deficient cells revealed H3K18 as a novel ubiquitination target of UHRF1 in mammalian cells. With bioinformatics and mutational analyses, we identified a ubiquitin interacting motif (UIM) in the N-terminal regulatory domain of DNMT1 that binds to ubiquitinated H3 tails and is essential for DNA methylation in vivo. H3 ubiquitination and subsequent DNA methylation required UHRF1 PHD binding to H3R2. These results show the manifold regulatory mechanisms controlling DNMT1 activity that require the reading and writing of epigenetic marks by UHRF1 and illustrate the multifaceted interplay between DNA and histone modifications. The identification and functional characterization of the DNMT1 UIM suggests a novel regulatory principle and we speculate that histone H2AK119 ubiquitination might also lead to UIM-dependent recruitment of DNMT1 and DNA methylation beyond classic maintenance.

UHRF1 as adaptor protein for DNA methylation and histone modification. UHRF1 interacts with proliferating cell nuclear antigen (PCNA, met 3.8%, Glu/Gln 2.87) at the replication fork and recognizes hemi-methylated CpG sites with subsequent recruitment of DNA (cytosine-5)-methyltransferase 1 (DNMT1) via its SRA domain for DNA methylation maintenance. Binding of ubiquitinated H3K18 (catalyzed by cooperative binding of TTD, PHD and SRA domains) to the ubiquitin interacting motif (UIM) present in the targeting sequence (TS) domain of DNMT1 is also essential for maintaining DNA methylation patterns. Recruitment of G9a methyltransferase to the SRA domain catalyzes di-and tri-methylation of H3 at lysine 9. Recognition of H3K9me3 by TTD and unmodified H3R2 by PHD further increases the chromatin binding ability of UHRF1. Further, histone deacetylase 1 (HDAC1: met 3.3%, Glu/Gln 3,2) binding to SRA domain ensures histone deacetylation which represses transcriptional process by promoting heterochromatin formation.

DEFINITION
A short protein description with the molecular wheight, isoforms, etc...
Use, when available, the link to Wikipedia (Es Trypsin)
External links not available on Wikipedia have to be added here
THE GENE
CHEMICAL STRUCTURE AND IMAGES
When relevant for the function
- Primary structure
- Secondary structure
- Tertiary structure
- Quaternary structure
h4. Protein Aminoacids Percentage (Width 700 px)


SYNTHESIS AND TURNOVER
mRNA synthesis
protein synthesis
post-translational modifications
degradation
CELLULAR FUNCTIONS
cellular localization,
biological function
- Cell signaling and Ligand transport
- Structural proteins
REGULATION
DIAGNOSTIC USE




PCNA is the latest protein of the machinery
It has 3.8% Met like mTOR that is concerned with high protein synthesis.
in this case probably high methylation rate
Combined blockade of AKT/mTOR pathway inhibits growth of human hemangioma via downregulation of proliferating cell nuclear antigen. 2012
- Abstract
Protein kinase B (AKT)/mammalian target of rapamycin (mTOR) signaling pathway plays a crucial role in the tumorigenesis and progression of multiple tumors, and has been shown to be important therapeutic targets for cancer. The present study aimed to explore the role and molecular mechanisms of AKT/mTOR pathway in human hemangioma (HA). Twenty-five cases of human HA tissues were collected. The expression of AKT, mTOR and proliferating cell nuclear antigen (PCNA) proteins was evaluated using semi-quantitative immunohistochemistry in biopsy samples in different phases of HA. AKT/mTOR pathway was blocked by recombinant small hairpin RNA adenovirus vector rAd5-AKT+mTOR (rAd5-Am), used for infecting proliferating phase HA-derived endothelial cells (HDEC). The expression of AKT, mTOR and PCNA was detected by Real-time PCR and Western blot assays. Cell proliferative activities were determined by MTT assay, and cell cycle distribution and apoptosis were analyzed by flow cytometry. As a consequence, the expression of AKT, mTOR and PCNA was significantly increased in proliferative phase HA, while that was decreased in involutive phase. Combined blockade of AKT/mTOR pathway by rAd5-Am diminished cell proliferative activities, and induced cell apoptosis and cycle arrest with the decreased expression of AKT, mTOR and PCNA in proliferative phase HDEC. In conclusion, the activity of AKT/mTOR pathway was increased in proliferative phase HA, while it was decreased in involutive phase. Combined blockade of AKT/mTOR pathway might suppress cell proliferation via down-regulation of PCNA expression, and induce apoptosis and cycle arrest in proliferative phase HDEC, suggesting that AKT/mTOR pathway might represent the important therapeutic targets for human HA.
CETN2 Met 5.8%
Un-healthy ageing and methylations in Paolo Vineis