- Origin
- What is scopolamine?
- Discovery
- Byosinthesis in plants
- Biological action
- Structure and molecular propriety
- Mechanism of action
- Effects
- Overdose
- Medical use
- Main pharmaceutic form
- Oftalmology
- Antidepressive
- Prevention and treatments postoperative nausea and vomiting
- Not medical use
- Police interrogations and "Truth Serum"
- Drug-facilited sexual assault
1 ORIGIN AND STRUCTURE
What is scopolamine?
The scopolamina, called also hyoscine, is an hallucinogenic alkaloid drug obtained from plants of the family Solenaceee, as Hyoscyamus niger or the species of the genus Datura; it's very abundant in the Datura Stramonium. Its anticholinergic properties give it some legitimate medical applications in very minute doses.
Datura Stramoium

Datura stramonium is a hallucinogenic plant, widely distributed and easily accessible, which contains toxic anticholinergic alkaloids. Voluntary or accidental ingestion can produce serious illness or death. The clinical presentation is similar to that seen in cases of atropine poisoning. Signs and symptoms such as mydriasis, tachycardia, arrhythmias, agitation, seizures and coma can appear. Diagnosis is clinical and early treatment should be given, including gastrointestinal decontamination, supportive care and physostigmine in life-threatening cases. Prognosis is usually favorable. Datura stramonium poisoning, July 2000
Discovery

The tropane alkaloids are among the oldest drugs known to man. The poisonous Solenacee family of plants, currently classified as genera: Atropa, Brugmansia, Datura, Duboisia, and Hyoscyamus Scopolia, with many species contain alkaloids, were well known in ancient times, and the relief of their use in folk medicine in different groups ethnic abundant. In 1819 Meissner (who coined the term alkaloid) was the first to realize that the active ingredients of these poisonous plants are alkaline and therefore can be isolated by extraction techniques, and consequently individual alkaloid compounds have begun to be isolated from 1830 onwards: atropine from Atropa belladona , and hyoscyamine from Hyoscyamus niger (K. Mein, PL Geiger, K. Hesse, 1831-1833), followed by scopolamine. Tropane alkaloids as medicinally useful natural products and their synthetic derivatives as new drugs, 2008
Scopolamine was discovered by A. Schmidt, of Marburg. Raehlmann was the first to give an account of its actions, based on clinical investigations. G. Gutmann, of Berlin, and L. Gross-mann, of Budapest, subsequently reported on its effects, also based on clinical experience in their respective clinics. As a member of the Solanaceae, it was a priori supposed to possess mydriatic qualities, and the investigations of different observers confirm this view.
(Grossman, Dr. Leopold. Therapeutische Monatshefte, Berlin, Marz, 1895. Gutmann, Dr. G. Ibid., 1894. Raehlmann, Prof. Dr. E. Ibid., Juli, 1894).
Biosynthesis in plants
The biosynthesis of scopolamine begins with the decarboxylation of L-ornithine to putrescine by ornithine decarboxylase . Putrescine is methylated to N-methylputrescine by putrescine N-methyltransferase. A putrescine oxidase that specifically recognizes methylated purtrescine catalizes the deamination of this compound to 4-methylaminobutanal which then undergoes a spontanous ring formation to N-Methyl-pyrrolium cation. In the next step, the pyrrolium cation condenses with acetoacetic acid yielding hygrine. No enzmyatic activity could be demonstrated that catalyzes this reaction. Hygrine further rearranges to tropinone. Subsequently, Tropinone reductase I converts tropinone to tropine which condenses with phenylalanine-derived phenyllactate to littorine. Alkaloid Biosynthesis: Metabolism and Trafficking, 2008

A cytochrome P450 classified as Cyp80F1 oxidizes and rearranges littorine to hyoscyamine aldehyde. In the final step, hyoscyamine undergoes epoxidation which is catalyzed by 6beta-hydroxyhyoscyamine epoxidase yielding scopolamine. Functional Genomic Analysis of Alkaloid Biosynthesis in Hyoscyamus niger Reveals a Cytochrome P450 Involved in Littorine Rearrangement, 2006
2 BIOLOGICAL ACTION
Structure and molecular propriety

The structure of scopolamine has been confirmed by total synthesis, which was achieved by several author. The stereocheminstry of scopolamine has been determined by chemical means as well as the X-ray crystalographic study of the hydro-bromide salt.
The structure of scopolamine hydrobromide revealed the confermation of the tropine residue to be as expected, with the six-membered ring in the chair conformation. The epoxide oxygen atom on the methylene bridge is in the boat configuration with respect to the nitrogen atom, and the methyl goup C(1) attached to the nitrogen atom is the axial position with respect to the six-membered ring.
The ester group attached to C(13) is in the axial position of the six-membered ring.
Molecular weight: 303.35 KDa
pH range: 4-5,5
(A complete review of the biochemistry of Scopolamine is here: Analytical Profiles of Drug Substances, by Klaus Flore
Mechanism of action
The alkaloid L-(-)-scopolamine [L-(-)-hyoscine] competitively inhibits muscarinic receptors for acetylcholine and acts as a nonselective muscarinic antagonist, producing both peripheral antimuscarinic properties and central sedative, antiemetic, and amnestic effects. The parasympatholytic scopolamine, structurally very similar to atropine (racemate of hyoscyamine), is used in conditions requiring decreased parasympathetic activity, primarily for its effect on the eye, gastrointestinal tract, heart, and salivary and bronchial secretion glands, and in special circumstances for a CNS action. (Pharmacokinetics and pharmacodynamics in clinical use of scopolamine, 2005)

Hyoscine (scopolamine) shows similar binding affinities to all of the five known muscarinic receptor sub-types. (Comparison of the effects of a selective muscarinic receptor antagonist and hyoscine (scopolamine) on motion sickness, skin conductance and heart rate, 1997)
Muscarinic acetylcholine receptors mediate diverse physiological functions. At present, five receptor subtypes (*M1 - M5*) have been identified. The odd-numbered receptors (M1, M3, and M5) are preferentially coupled to Gq/11 and activate phospholipase C, which initiates the phosphatidylinositol trisphosphate cascade leading to intracellular Ca2+ mobilization and activation of protein kinase C. On the other hand, the even-numbered receptors (M2 and M4) are coupled to Gi/o, and inhibit adenylyl cyclase activity. They also activate G protein-gated potassium channels, which leads to hyperpolarization of the plasma membrane in different excitable cells. Individual members of the family are expressed in an overlapping fashion in various tissues and cell types. Recent gene targeting approaches have unraveled the specific function of these muscarinic receptor subtypes, which were not able to be fully elucidated with pharmacological approaches because of the non-selective effects of the available ligands. Based on these findings, muscarinic receptors have been emerging as an important therapeutic target for various diseases, including dry mouth, incontinence and chronic obstructive pulmonary disease.
(Muscarinic Acetylcholine Receptors, by Masaru Ishii, Yoshihisa Kurachi)

Effects
There are two main types of cholinergic receptors widely distributed throughout the brain. These receptors are classified as muscarinic and nicotinic receptors. In certain regions of the brain only the muscarinic subtype is found eg midbrain, medulla, and pons while in other regions eg substantia nigra, locus coeruleus and septum only the nicotinic receptor subtype is found. Both sub-types are located in the corpus striatum, cerebral cortex, hippocampus, thalamus, hypothalamus and cerebellum.

Scopolamine is a very potent psychoactive drug that is used as a standard/reference drug for inducing amnesia in mammals. A characteristic feature of these alkaloids is that subjects do not recall memories of the time they were intoxicated, and the user loses all sense of reality. The effects are generally interpreted as a cholinergic deficit and related to the fact that acetylcholine is involved in memory functions. The use of scopolamine as a pharmacological model of ‘cholinergic amnesia’ became very popular after the cholinergic hypothesis of geriatric memory dysfunction was postulated. (The cholinergic hypothesis of geriatric memory dysfunction, 1982)
This hypothesis assumes that the age-related decline in cognitive functions is predominantly related to the decrease of the integrity of cholinergic neurotransmission. Since scopolamine-induced amnesia was proposed to be due to blockade of cholinergic neurotransmission, this substance was used to model the cognitive deficits that could be observed in aging and dementia. Scopolamine appears to be a nonselective muscarinic receptor antagonist and it has been demonstrated that scopolamine has a high selectivity for the muscarinic receptor (The Binding of Antagonists to Brain Muscarinic Receptors, 1978), although it has been reported that high doses of scopolamine are also blocking nicotinic receptors. ( Binding of tropane alkaloids to nicotinic and muscarinic acetylcholine receptors, 1995).
Scopolamine acts as a nonselective muscarinic antagonist, producing both peripheral antimuscarinic properties and central sedative, antiemetic, and amnestic effects. The parasympatholytic scopolamine, structurally very similar to atropine (racemate of hyoscyamine), is used in conditions requiring decreased parasympathetic activity, primarily for its effect on the eye, gastrointestinal tract, heart, and salivary and bronchial secretion glands, and in special circumstances for a CNS action. Therefore, scopolamine is most suitable for premedication before anesthesia and for antiemetic effects. This alkaloid is the most effective single agent to prevent motion sickness. Scopolamine was the first drug to be made commercially available in a transdermal therapeutic system (TTS-patch) delivering alkaloid. (Scopolamine (hyoscine) for preventing and treating motion sickness, 2007).
Distribution of main muscarinic receptors

Overdose
In acute overdosage, the main clinical problem is central nervous system (CNS) depression. In Australia, tablets containing scopolamine hydrobromide 0.3 mg are available over the counter in packs of ten. The recommended dose for adults is one to two tablets as a single dose, repeated four to six hours later, if required. The maximum dose stated on the pack is four tablets over a 24-hour period with a caution regarding drowsiness and blurred vision. In the study reported below there is a patient who presented with symptoms of anticholinergic syndrome secondary to an unintentional overdose of scopolamine. Whilst at work, the patient noticed that he had forgotten his prescribed medication, domperidone, at home; a friend gave him some travel sickness medication which contained scopolamine for relief of nausea. On a previous occasion, he had experienced a similar, less severe reaction with another anticholinergic agent, loperamide. This report highlights the need to consider nonprescription products, ie, over the counter medications, herbal/nutritional supplements as causes of anticholinergic syndrome when a patient presents with symptoms suggestive of this diagnosis. (Anticholinergic syndrome following an unintentional overdose of scopolamine, 2009).

Overdosage also cause deteriorated cognitive function and changed mental status acutely. Besides flushing skin and psychotic behaviors including bizarre actions, hallucinations, aggressive behavior, hyperactivity, and incoherent speech are also noticed. Symptoms and signs are resolved after removal of scopolamine patch and conservative management. (Delirium due to scopolamine patch in a 4-year-old boy, 2011).

Scopolamine was a suspect carcinogen because it contains an aliphatic epoxide moiety which may act as a biological alkylating agent. (NTP Toxicology and Carcinogenesis Studies of Scopolamine Hydrobromide Trihydrate (CAS No. 6533-68-2) in F344 Rats and B6C3F1 Mice (Gavage Studies), 2007).
A complete review about scopolamine overdose:
#D.K. Van Sassenbroeck, D.M. Hemelsoet, P. Vanwalleghem, A.G. Verstraete, P. Santens, K.G. Monsieurs, and W.A. Buylaert. Three cases of substitution errors leading to hyoscine hydrobromide overdose. Clin. Toxicol. 43: 861–865 (2005). Link here
#M. Balikova. Collective poisoning with hallucinogenous herbal tea. Forensic Sci. Int. 128: 50–52 (2002). Link here
#S.W. Cheng, W.H. Hu, D.Z. Hung, and D.Y. Yang. Anticholinergic poisoning from a large dose of scopolia extract. Vet. Hum. Toxicol. 44: 222–223 (2002). Link here.
#A. Ardila and C. Moreno. Scopolamine intoxication as a model of transiet global amnesia. Brain Cogn. 15: 236–245 (1991). Link here.
3 MEDICAL USE
Pharmaceutic form (Buscopan)
The N -butyl bromide derivative of scopolamine, available commercially as Buscopan®, is commonly used as an antispasmotic.
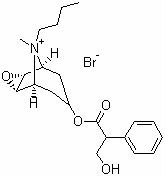
Buscopan, which contains N-butyl scopolammonium bromide (BSB), is indicated for the relief of abdominal discomfort, pain, and acute colic. As a quaternary ammonium compound with low lipid solubility, BSB cannot pass the blood-brain barrier easily and only rarely causes the central nervous system side effects associated with atropine and scopolamine. (Buscopan for oesophageal food bolus impaction, 2007).

Abdominal cramping and pain is a frequent problem in the adult population of Western countries, with an estimated prevalence of < or =30%. Hyoscine butylbromide (scopolamine butylbromide) [Buscopan/Buscapina] is an antispasmodic drug indicated for the treatment of abdominal pain associated with cramps induced by gastrointestinal (GI) spasms. It was first registered in Germany in 1951 and marketed in 1952, and has since become available worldwide both as a prescription drug and as an over-the-counter medicine in many countries. Pharmacological studies have revealed that hyoscine butylbromide is an anticholinergic drug with high affinity for muscarinic receptors located on the smooth-muscle cells of the GI tract. Its anticholinergic action exerts a smooth-muscle relaxing/spasmolytic effect. Blockade of the muscarinic receptors in the GI tract is the basis for its use in the treatment of abdominal pain secondary to cramping. Hyoscine butylbromide also binds to nicotinic receptors, which induces a ganglion-blocking effect. Because of its high tissue affinity for muscarinic receptors, hyoscine butylbromide remains available at the site of action in the intestine and exerts a local spasmolytic effect. (Hyoscine butylbromide: a review of its use in the treatment of abdominal cramping and pain, 2007).
Oftalmology (Isopto Hyoscine)

Scopolamine ophthalmic (Isopto Hyoscine) causes the muscles in the eye to become relaxed: this widens the pupil, which will remain wide and will not respond to light.
Scopolamine ophthalmic is used to dilate (widen) pupil when there is an inflammatory condition or in diagnostic or postsurgery situations in which this effect may be helpful.
(Ocular effects of scopolamine dermal patch in open-angle glaucoma, 2004).
Antidepressive
Hypersensitivity of the cholinergic system plays a central role in the pathogenesis of mood disorders. The demonstration that an antimuscarinic agent, as Scopolamine, produces potent antidepressant effects provides a strong link between muscarinic receptor function and mood disorders. (Antidepressant efficacy of the antimuscarinic drug scopolamine: a randomized, placebo-controlled clinical trial, 2006).
Prevention and treatments postoperative nausea and vomiting
Transdermal scopolamine (TDS) is a centrally acting anticholinergic agent that was approved in 2001 by the US Food and Drug Administration for the prevention of postoperative nausea and vomiting (PONV). Although TDS has been reported to be clinically efficacious in the prevention of PONV, several adverse events (AEs), such as sedation, dry mouth, blurred vision, central cholinergic syndrome, and confusion (particularly in elderly patients with mild cognitive impairment), are potential concerns.
(Transdermal scopolamine for the prevention of postoperative nausea and vomiting: a systematic review and meta-analysis, 2010).
4 NOT MEDICAL USE
Police interrogations and "Truth Serum"

Early in this century physicians began to employ scopolamine, along with morphine and chloroform, to induce a state of "twilight sleep" during childbirth. A constituent of henbane, scopolamine was known to produce sedation and drowsiness, confusion and disorientation, incoordination, and amnesia for events experienced during intoxication. Yet physicians noted that women in twilight sleep answered questions accurately and often volunteered exceedingly candid remarks.
In 1922 it occurred to Robert House, a Dallas, Texas, obstetrician, that a similar technique might be employed in the interrogation of suspected criminals, and he arranged to interview under scopolamine two prisoners in the Dallas county jail whose guilt seemed clearly confirmed. Under the drug, both men denied the charges on which they were held; and both, upon trial, were found not guilty. Enthusiastic at this success, House concluded that a patient under the influence of scopolamine "cannot create a lie ... and there is no power to think or reason." His experiment and this conclusion attracted wide attention, and the idea of a "truth" drug was thus launched upon the public consciousness.
The phrase "truth serum" is believed to have appeared first in a news report of House's experiment in the Los Angeles Record, sometime in 1922. House resisted the term for a while but eventually came to employ it regularly himself. He published some eleven articles on scopolamine in the years 1921-1929, with a noticeable increase in polemical zeal as time went on. What had begun as something of a scientific statement turned finally into a dedicated crusade by the "father of truth serum" on behalf of his offspring, wherein he was "grossly indulgent of its wayward behavior and stubbornly proud of its minor achievements."
Only a handful of cases in which scopolamine was used for police interrogation came to public notice, though there is evidence suggesting that some police forces may have used it extensively. One police writer claims that the threat of scopolamine interrogation has been effective in extracting confessions from criminal suspects, who are told they will first be rendered unconscious by chloral hydrate placed covertly in their coffee or drinking water.
Because of a number of undesirable side effects, scopolamine was shortly disqualified as a "truth" drug. Among the most disabling of the side effects are hallucinations, disturbed perception, somnolence, and physiological phenomena such as headache, rapid heart, and blurred vision, which distract the subject from the central purpose of the interview. Furthermore, the physical action is long, far outlasting the psychological effects. Scopolomine continues, in some cases, to make anesthesia and surgery safer by drying the mouth and throat and reducing secretions that might obstruct the air passages. But the fantastically, almost painfully, dry "desert" mouth brought on by the drug is hardly conducive to free talking, even in a tractable subject.
(Truth Drugs in Interrogation, 1993).
Drug-facilited sexual assault

Place that may lead to impairment of consciousness followed by amnesia, scopolamine is sometimes administered dissolved in beverages unsuspecting victim to commit robbery or sexual assault. (Analytical developments in toxicological investigation of drug-facilitated sexual assault, 2003).