
Peppermint oil is extracted from mentha x piperita that is a cross between watermint and spearmint and is native to Europe. Historically, the herb has been known for its medicinal uses, and its impressively long history often gives it the prestigious title as the world’s oldest medicine (peppermint has a long tradition of medicinal use, with archaeological evidence placing its use as at ten thousand years ago, or longer).
Uses
The health benefits of peppermint oil include its ability to treat indigestion, respiratory problems, headache, nausea, fever, stomach and bowel spasms, as well as for pain relief. The aroma of peppermint has been found to enhance memory and alertness, although other research contests this. Peppermint is used in aromatherapy. Due to the presence of menthol, menthone and menthyl esters, peppermint and peppermint oil find wide applications in the manufacturing of soap, shampoo, cigarettes, toothpaste, chewing gum, tea and ice cream as a flavoring agent.

Health Benefits of Peppermint Oil
Chemical constituents
Peppermint has a high menthol content. The oil also contains menthone and menthyl esters, particularly menthyl acetate. Dried peppermint typically has 0.3-0.4% of volatile oil containing menthol (7-48%), menthone (20-46%), menthyl acetate (3-10%), menthofuran (1-17%) and 1,8-cineol (3-6%). Peppermint oil also contains small amounts of many additional compounds including limonene, pulegone (a natural pesticide), caryophyllene and pinene.
Menthol
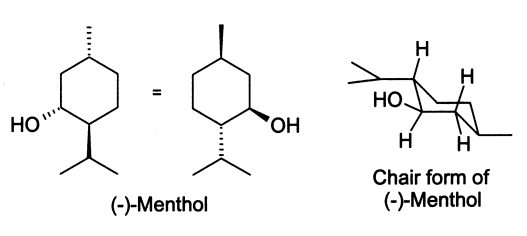
Menthol is a waxy, crystalline substance, clear or white in color, which is solid at room temperature and melts slightly above. The main form of menthol occurring in nature is (-)-menthol, which is assigned the (1R,2S,5R) configuration. Menthol gives peppermint oil its local anesthetic and analgesic qualities through the activation of cold-sensitive TRPM8 receptors in the skin and mucosal tissues, that are the primary source of the cooling sensation that follows the topical application of peppermint oil. In this sense, it is similar to capsaicin, the chemical responsible for the spiciness of hot chilis (which stimulates heat sensors, also without causing an actual change in temperature).
Menthol: a refreshing look at this ancient compound
Menthol: a simple monoterpene with remarkable biological properties
Menthol
Menthol and Smoking Dependence
Mechanism of action on skin and mucosal tissue
Peppermint oil, trough its main component (menthol), activates the transient receptor potential melastatin 8 (TRPM8) channel, also known as the cold and menthol receptor 1 (CMR1), is a protein that in humans is encoded by the TRPM8 gene.
Transient Receptor Potential Melastin 8 Channel Structure
The TRPM8 channel is a homotetramer, composed of four identical subunits with a transmembrane domain with six helices (S1-S6). The first four, S1-S4, act as the voltage sensor and allow binding of menthol, icilin and similar channel agonists. S5 and S6 and a connecting loop, also part of the structure, make up the pore, a non-selective cation channel which consists of a highly conserved hydrophobic region. A range of diverse components are required for the high level of specificity in responding to result in ion flow to cold and menthol stimuli.

Function
TRPM8 is an ion channel, upon activation it allows the entry of Na + (sodium) and Ca 2+ (calcium) ions to the cell that leads to depolarization and the generation of an action potential. The signal is conducted from primary afferents (type C- and A-delta) eventually leading to the sensation of cold and cold pain.
The TRPM8 protein is expressed in sensory neurons, and it is activated by cold temperatures and cooling agents, such as menthol and icilin that are the most selective agonist of TRPM8.
TRPM8 is also expressed in the prostate, lungs, and bladder where its function is not well understood.
The transient receptor potential channel (TRPs) superfamily, which includes the menthol (TRPM8) and capsaicin receptors (TRPV1), serve a variety of functions in the peripheral and central nervous systems. In the peripheral nervous system, TRPs respond to stimuli from temperature, pressure, inflammatory agents, and receptor activation. Central nervous system roles of the receptors include neurite outgrowth, receptor signaling, and excitoxic cell death resulting from noxious stimuli.
McKemy et al., 2002 provided some of the first evidence for existence of a cold-activated receptor throughout the mammalian somatosensory system. Using calcium imaging and patch clamp based approaches, they showed a response in dorsal root ganglion (DRG) neurons that exposure to cold, 20 °C or cooler, lead to a response in calcium influx. This receptor was shown to respond to both cold temperatures, menthol, and similar now-known agonists of the TRPM8 receptor. Application of menthol to skin or mucus membranes results directly in membrane depolarization, followed by calcium influx via voltage-dependent calcium channels, providing evidence for the role of TRPM8 and other TRP receptors to mediate our sensory interaction with the environment in response to cold in the same way as in response to menthol.
Pharmacology of an important naturally medicinal cool
Modulation of TRPM8 in Sensory Neurons
Several cellular signaling cascades may be involved in the regulation of TRPM8 activity in sensory neurons. Activation of TRPM8 by physical (cold) or chemical (menthol) stimulation is followed by channel desensitization, which depends on extracellular Ca 2+, and there is evidence for an involvement of phosphatidylinositol 4,5-biphosphate (PIP 2) in this process. PIP 2 acts as a positive modulator of cold or menthol sensitivity of TRPM8, most likely by shifting the voltage-sensitivity of activation toward physiological voltages, and prevents current rundown in cell-free patches. It has been proposed that Ca 2+ influx through TRPM8 leads to activation of Ca 2+-dependent phospholipase C, inducing a reduction of PIP 2 levels and channel desensitization. A complementary pathway for TRPM8 desensitization may occur via activation of Ca 2+-dependent protein kinase C (PKC), which indirectly causes dephosphorylation of TRPM8 via protein phosphatase 1, leading to downregulation of the channel. The sensitivity of TRPM8 to PKC and PIP 2 suggests that in vivo TRPM8 could be highly sensitive to stimulation of phospholipase C (PLC)-coupled receptors. Activation of PLC would have a dual inhibitory effect on the channel via a reduction of cellular PIP 2 levels and via a diacyl glycerol-induced activation of PKC. Recently, Daniels et al. showed evidence supporting the notion that PLC activity mediates adaptation of TRPM8 to thermal stimuli.
Bradykinin and prostaglandin E2, two pro-inflammatory mediators, applied acutely reduce the responses to cold and menthol in putative TRPM8-positive neurons, possibly by PKC and protein kinase A-dependent mechanisms, respectively. Phospholipase A2 (PLA2) could also affect the function of TRPM8, by generating polyunsaturated fatty acids (PUFAs) and lysophospholipids from glycerophospholipids that modulate the channel in opposite directions. On the other hand, Gi-coupled α 2A -adrenoreceptor (α 2A -AR), which is also expressed in sensory neurons, could modulate the function of TRPM8; in DRG neurons, stimulation of α 2A -AR reduces the activity of the channel by a PKA-dependent phosphorylation mechanism

A Cool Channel in Cold Transduction
Conclusion
Cold-patches and peppermint oil have traditionally been used to induce analgesia or relief in pain which is caused as result of traumatic injuries but the underlying mechanism of cold-induced analgesia remained obscure until the discovery of TRPM8 (in 2001). Although the use of peppermint oil in traditional medicine is millenary, only recently it has been possible to start unravelling the secrets of this versatile essential oil.