DEFINITION
The ichthyoses are characterized clinical for the generalized desquamation. The name ichthyosis derives from the Greek ichthys that it means “fish” and one refers to the likeness of the cute to the squame of the fish. The ichthyoses represent a heterogenous group of diseases, with hereditary and acquired forms.
(Fitzpatrick’s, Dermatology in General Medicine, Fifth Edition)
Harlequin type ichthyosis, the most severe form of congenital ichthyosis, is characterized by a thickening of the keratin layer in fetal skin. The afflicted child is born with not skin, but massive, diamond-shaped scales. As well, the eyes, ears, mouth, and other appendages can be abnormally contracted. The scaly armor limits the child’s movement, and because it is cracked where normal skin would fold, it is useless for skin’s primary function – protection. Bacteria and other contaminants easily pass into the cracks and can cause lethal infections.
The term harlequin comes from both the baby’s facial expression and the diamond-shaped pattern of the scales. (Known as hyperkeratosis) 17th century entertainers known as jesters, or harlequins, wore costumes with diamond patterns on them, as well as a particular style of face paint.
The disease is also known as harlequin ichthyosis, ichthyosis congenita, and keratosis diffusa fetalis. Sufferers are known as harlequin fetuses, harlequin babies, or plain harlequins.


EPIDEMIOLOGY
Frequency
International. More than 100 cases of harlequin ichthyosis have been reported.
Mortality/Morbidity
The mortality for harlequin ichthyosis rate is high. With neonatal intensive care and the advent of retinoid therapy, some babies have survived the newborn period. They are still at risk of dying from systemic infection, which is the most common cause of death.
Race
No racial predilection is known for harlequin ichthyosis.
Sex
No increased risk of harlequin ichthyosis based on sex is known.
SYMPTOMS
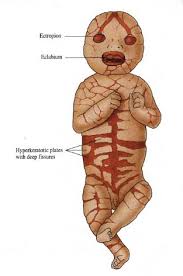
•Skin: Severely thickened skin with large, shiny plates of hyperkeratotic scale is present at birth. Deep, erythematous fissures separate the scales.
•Eyes: Severe ectropion is present. The free edges of the upper and lower eyelids are everted, leaving the conjunctivae at risk for desiccation and trauma.
•Ears: The pinnae may be small and rudimentary or absent.
•Lips: Severe traction on the lips causes eclabium and a fixed, open mouth. This may result in feeding difficulties.
•Nose: Nasal hypoplasia and eroded nasal alae may occur.
•Extremities: The limbs are encased in the thick, hyperkeratotic skin, resulting in flexion contractures of the arms, the legs, and the digits. Limb motility is poor to absent. Circumferential constriction of a limb can occur, leading to distal swelling or even gangrene. Hypoplasia of the fingers, toes, and fingernails is reported. Polydactyly is described.
•Temperature dysregulation: Thickened skin prevents normal sweat gland function and heat loss. The infants are heat intolerant and can become hyperthermic.
•Respiratory status: Restriction of chest-wall expansion can result in respiratory distress, hypoventilation, and respiratory failure.
•Hydration status: Dehydration from excess water loss can cause tachycardia and poor urine output.
•Central nervous system: Metabolic abnormalities can cause seizures. CNS depression can be a sign of sepsis or hypoxia. Hyperkeratosis may restrict spontaneous movements, making neurologic assessment difficult.

DIAGNOSIS
Up to now the preborn diagnosis them was based on the biopsy of fetal skin carried out in an advanced phase of the pregnancy. (In past the fetal biopsy was executed through the fetoscopy and it had a mortality rate that was between 4-7%. Today it is executed under echographyc guide between 17a and 20the week of gestation, with a biopsy clamp. The samples should be aimed at the zone of cute affected by the eventual dermatological lesion. You have to introduce the needle and then the biopsy clamp: the teeth of the clamp are opened, it is pushed out on the cute fetal and the sample is carried out. The diagnosis is made with a conventional histological examination and with the electron microscope, analyzing the structure and the ultrastructure of the cute and resorting to the immunohistochemical analyses. The most important indication to the fetal cutaneous biopsy is the diagnosis of the genodermatosis, hereditary diseases of the skin with high morbidity and mortality, including Harlequin ichthyosis.). It is predictable that for the Harlequin Ichthyosis the preborn test based on DNA will replace the fetal biopsy. This analysis may be made before the tenth week of gestation by a normal chorionic villus sampling, or even before with the non-invasive analysis of the DNA of fetal cells in maternal circulation. Finally, knowledge of the exact mutations (gene ABCA12) in this serious disease is the basis for the subsequent development of tools for preimplantation genetic diagnosis [assisted reproductive technology that allows carriers to select healthy embryos].
PATHOGENESIS
Genetic factors
Mutations in a gene known as ABCA12 (adenosine triphosphate [ATP]-binding cassette transporter, subfamily A, member 12), in chromosome region 2q35, underlie this disorder.

Patients with harlequin ichthyosis are usually homozygous for this mutation consistent with autosomal recessive inheritance.
The ABC superfamily of genes encodes proteins that transport a number of substrates across cell membranes. ABCA12 is thought to encode a transmembrane protein that mediates lipid transport.
This ABCA12 -mediated lipid-transfer system is thought to be essential to the transfer of lipids from the cytosol of the corneocyte into lamellar granules. Lamellar granules are intracellular granules that originate from the Golgi apparatus of keratinocytes in the stratum corneum. These granules are responsible for secreting lipids that maintain the skin barrier at the interface between the granular cell layer and the cornified layer. The extruded lipids are arranged into lamellae in the intercellular space with the help of concomitantly released hydrolytic enzymes. The lamellae form the skin’s hydrophobic sphingolipid seal.
In harlequin ichthyosis, the ABCA12 -mediated transfer of lipid to lamellar granules is absent. The lamellar granules themselves are morphologically abnormal or absent. Normal extrusion of lipid from these granules into the intercellular space cannot occur, and lipid lamellae are not formed. This defective lipid “mortar” between corneocyte “bricks” results in aberrant skin permeability and lack of normal corneocyte desquamation.
The exact mechanism of this transport abnormality has yet to be elucidated. One hypothesis involves abnormal calcium-mediated signaling by means of calpains. Calpains are calcium-activated neutral proteases that are essential to normal epidermal differentiation. Calpains are consistently underexpressed in patients with harlequin ichthyosis compared with the general population.
The pivotal role of ABCA12 in harlequin ichthyosis is supported by in vitro data. Studies have demonstrated normalization of lipid transport when the wild-type ABCA12 gene is transferred to keratinocytes of patients with harlequin ichthyosis.
A milder form of ichthyosis, lamellar ichthyosis type 2, also involves mutations in the ABCA12 gene. The phenotypic difference between the disorders has been explained on the basis of differing genotypic variants. Nonsense mutations in ABCA12 are seen in harlequin ichthyosis, whereas missense mutations underlie lamellar ichthyosis type 2.
Other chromosomal abnormalities are described. One patient with a de novo deletion of chromosome arm 18q has been reported.
Histopathologic, ultrastructural, and biochemical factors
Histopathologic, ultrastructural, and biochemical studies have identified several characteristic abnormalities in the skin of patients with harlequin ichthyosis.
The 2 main abnormalities involve lamellar granules and the structural proteins of the cell cytoskeleton. The relationship between mutations in the gene ABCA12 and abnormal lamellar granules is well documented. The pathophysiology of the other abnormalities documented below has yet to be elucidated.
Abnormal lamellar granule structure and function
The pivotal role of lamellar granules in maintaining a normal skin barrier is described in the Genetic factors bullet points at the beginning of this section.
All patients with harlequin ichthyosis have absent or defective lamellar granules and no intercellular lipid lamellae. The lipid abnormality is believed to allow excessive transepidermal water loss. Lack of released hydrolases prevents desquamation, resulting in a severe retention hyperkeratosis.
Abnormal conversion of profilaggrin to filaggrin
Profilaggrin is a phosphorylated polyprotein residing in keratohyalin granules in keratinocytes in the granular cell layer. During the evolution to the corneal layer, profilaggrin converts to filaggrin by means of dephosphorylation. Filaggrin allows dense packing of keratin filaments. Its subsequent breakdown into amino acids occurs prior to desquamation of the stratum corneum.
Some patients have a persistence of profilaggrin and an absence of filaggrin in the stratum corneum. A defect in protein phosphatase activity and subsequent lack of conversion of profilaggrin to filaggrin is hypothesized.
Abnormal expression of keratin
Keratinocyte cell cultures have yielded interesting and heterogeneous findings among patients with harlequin ichthyosis.
Keratin filament density is low in most patients. Expression of certain keratins is abnormal in some patients and normal in others. How this altered expression of structural proteins influences desquamation is uncertain.
Abnormal keratohyalin granules
Keratohyalin granules are identified by antifilaggrin antibodies and can be abnormal in some patients with harlequin ichthyosis. They can be large and stellate, small and rounded, or absent.
THERAPY
Treatment with isotretinoin or etretinate during the neonatal period may assist in the scaling of the membrane. Advances in neonatal intensive care along with the scaling made easier by the use of systemic retinoids has led to improved survival and use of the name ‘harlequin baby’ instead of the name ‘Harlequin fetus’.
The baby may require intensive care monitoring of fluid and electrolytes. Specific treatments of the skin can minimize the formation of vesicles and increase the healing of erosions to decrease the friction and mechanical trauma should be used in lubricants, protective padding and special bandages. The baby is prone to erosion with large bacterial infections and sepsis, the use of topical and systemic antibiotics chosen with care, can minimize the extent of infection.