An antibody (Ab), also known as an immunoglobulin, is a large, Y-shaped protein produced mainly by plasma cells that is used by the immune system to neutralize pathogens such as pathogenic bacteria and viruses.
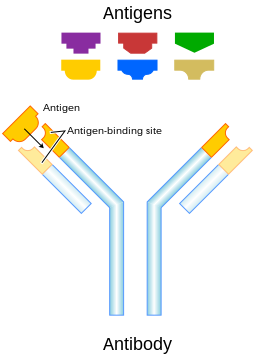
The antibody recognizes a unique molecule of the pathogen, called an antigen, via the Fab's variable region.[3] Each tip of the "Y" of an antibody contains a paratope (analogous to a lock) that is specific for one particular epitope (similarly analogous to a key) on an antigen, allowing these two structures to bind together with precision. Using this binding mechanism, an antibody can tag a microbe or an infected cell for attack by other parts of the immune system, or can neutralize its target directly (for example, by blocking a part of a microbe that is essential for its invasion and survival). Depending on the antigen, the binding may impede the biological process causing the disease or may activate macrophages to destroy the foreign substance. The ability of an antibody to communicate with the other components of the immune system is mediated via its Fc region (located at the base of the "Y"), which contains a conserved glycosylation site involved in these interactions. The production of antibodies is the main function of the humoral immune system.
the Fc Receptor

An Fc receptor is a protein found on the surface of certain cells – including, among others, B lymphocytes, follicular dendritic cells, natural killer cells, macrophages, neutrophils, eosinophils, basophils, human platelets, and mast cells – that contribute to the protective functions of the immune system. Its name is derived from its binding specificity for a part of an antibody known as the Fc (Fragment, crystallizable) region. Fc receptors bind to antibodies that are attached to infected cells or invading pathogens. Their activity stimulates phagocytic or cytotoxic cells to destroy microbes, or infected cells by antibody-mediated phagocytosis or antibody-dependent cell-mediated cytotoxicity.
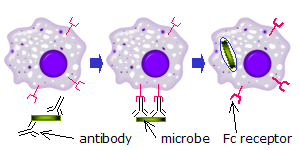
When IgG molecules, specific for a certain antigen or surface component, bind to the pathogen with their Fab region (fragment antigen binding region), their Fc regions point outwards, in direct reach of phagocytes. Phagocytes bind those Fc regions with their Fc receptors. Many low affinity interactions are formed between receptor and antibody that work together to tightly bind the antibody-coated microbe. The low individual affinity prevents Fc receptors from binding antibodies in the absence of antigen, and therefore reduces the chance of immune cell activation in the absence of infection. This also prevents agglutination (clotting) of phagocytes by antibody when there is no antigen. After a pathogen has been bound, interactions between the Fc region of the antibody and the Fc receptors of the phagocyte results in the initiation of phagocytosis.

Lysosomes are membrane-enclosed acid vesicles that contain an array of enzymes capable of breaking down all types of biological polymers—proteins, nucleic acids, carbohydrates, and lipids. Lysosomes function as the digestive system of the cell, serving both to degrade material taken up from outside the cell and to digest obsolete components of the cell itself. In their simplest form, lysosomes are visualized as dense spherical vacuoles, but they can display considerable variation in size and shape as a result of differences in the materials that have been taken up for digestion. Lysosomes thus represent morphologically diverse organelles defined by the common function of degrading intracellular material.
On the basis of this description it is quite clear that phagocytosis of infectious agents is simply an instance of the more general mechanism of cell feeding.