Neuroactive steroids modify neuronal excitability through interaction with neurotransmitter-gated ion channels (non genomic effect). In addition, these steroids may also exert effects on gene expression via intracellular steroid hormone receptors (genomic effect). Many of the enzymes involved in their metabolism are shared by adrenals steroids metabolism.

Neurosteroids and GABAA receptor interactions: a focus on stress, 2011
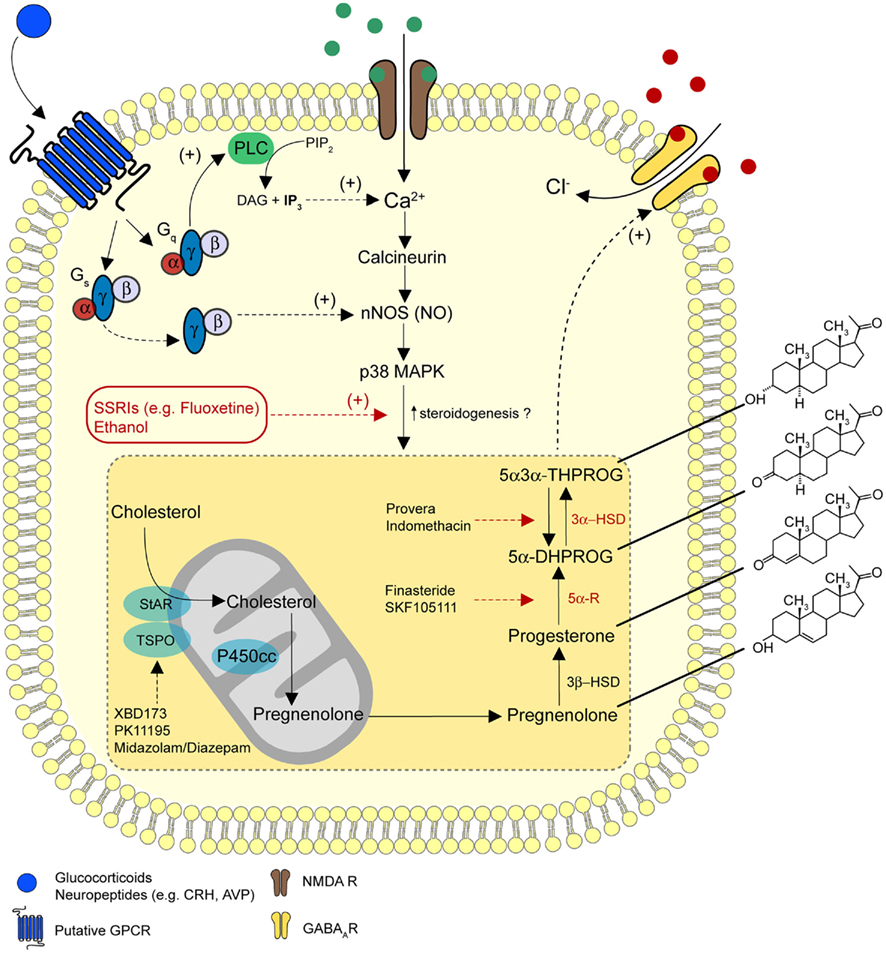
- The chemical structure of endogenous 5alpha-pregnan-3alpha,21-diol-20-one (3alpha,5alpha-THDOC) and synthetic (alphaxalone, ganaxolone and 3alpha,5alpha-17-phenylandrost-16-en-3-ol, or 17PA) steroids. b | Biosynthesis of GABA (gamma-aminobutyric acid)-modulatory neurosteroids. The pathway for the synthesis of 5alpha-pregnan-3alpha-ol-20-one (3alpha,5alpha-THPROG) from cholesterol is shown. Also indicated is the site of action of drugs that have been used to evaluate the influence of endogenous 3alpha,5alpha-THPROG on inhibitory neurotransmission. Steroidogenic acute-regulatory protein (StAR) might interact with the mitochondrial benzodiazepine receptor (MBR) to facilitate the transport of cholesterol across the mitochondrial membrane. 3alpha-HSD, 3alpha-hydroxysteroid dehydrogenase; 3beta-HSD, 3beta-hydroxysteroid dehydrogenase; 5alpha-DHPROG, 5alpha-dihydroprogesterone; P450scc, P450 side-chain cleavage.
Neurosteroids: endogenous regulators of the GABAA receptor 2005

Steroids
more neurosteroids
Human steroid and oxysterol 7α-hydroxylase CYP7B1: substrate specificity, azole binding and misfolding of clinically relevant mutants, 2014
- The characteristic function of CYP7B1, in contrast to cholesterol 7α-hydroxylase (CYP7A1), is in the metabolism of neurosteroids. Neurosteroids are synthesized de novo in the central nervous system from cholesterol [32, 33] by steroidogenic cytochrome P450s and steroid dehydrogenases, i.e. cholesterol side chain cleavage enzyme (P450scc, CYP11A1), oxysterol 7α-hydroxylase (CYP7B1), 3β-hydroxysteroid dehydrogenase/Δ5,Δ4-isomerase (3β-HSD), steroid 17α-hydroxylase/17,20-lyase (CYP17) and 17β-HSD. PREG and DHEA are well known for their neuroprotective properties and ability to enhance memory [34]. The product of PREG conversion by CYP7B1, 7α-hydroxy-PREG, is also classified as a neurosteroid [35]. Its physiological function is more clearly understood in birds, newts [36] and rats [37] than in humans. The 7α-hydroxy-PREG mediates melatonin action on diurnal locomotor rhythms in quail [38], acts as a neuronal activator to stimulate locomotor activity via the dopaminergic system in breeding newts [39, 40] and improves spatial memory retention in cognitively impaired aged rats [37]. These studies show that 7-hydroxy-PREG is a more potent neurosteroid than PREG, thus indicating an activating function of CYP7B1, which is consistent with the low activity of CYP7B1 toward this steroid (Table 1). Another CYP7B1 product, 7α-hydroxy-EpiA, has been reported to reduce ischemia-induced neuronal damage [41]. However, the nature of such neuroprotective properties is unknown.
- The role of the 7α-hydroxylated product of DHEA is less studied but has been suggested to have immunomodulatory and antiglucocorticoid effects similar to DHEA [42]. Interestingly, both products of CYP7B1, 7α-hydroxy-PREG and 7α-hydroxyDHEA, may exert antiglucocorticoid effects in target tissues by competing with 11-ketoglucocorticoids for access to 11β-HSD1, thus attenuating regeneration of active glucocorticoids [43]. The 11β-HSD1 has also been shown to carry out the interconversion of 7α-hydroxy-DHEA into 7β-hydroxy-DHEA, at least in vitro [18]. The same localization of 11β-HSD1 and CYP7B1 in the hippocampus, on one hand, and the rotational symmetry between the 7α- and 11β-positions of the steroid nucleus, on the other hand, suggest a functional overlap between glucocorticoid and neurosteroid metabolism.
Enzymes
Steroid-5-alpha-reductase
Steroid-5-alpha-reductase and autism?
AKR1C4/3alpha-HSD belongs lo the aldo/keto reductase superfamily
Mechanism of Action
Neurosteroids are produced by both neuron and glial cells.

It is usually associated with GABA synthesis

Synthesis regulation

Alcohol withdrawal syndrome: mechanisms, manifestations, and management, 2016
Update on the Neurobiology of Alcohol Withdrawal Seizures, 2005
Drugs
Agonists
Antagonists
Diversity of Steroid Hormone Actions on the Brain
Loss of neurosteroid-mediated protection following stress during fetal life