THE DEADLY STING OF NANOBEES
Molecularly targeted nanocarriers deliver the cytolytic-peptide, melittin, specifically to tumor cells in mice, reducing tumor growth, 2009
Experimental details
What is Melittin?
Melittin structure
Melittin is is the principal active component of apitoxin (52% of bee venom); is a water-soluble, cationic 26-residue peptide, in which residues 1±20 (N-terminal) form an amphipathic helical segment and residues 21±26 (C-terminal) constitute a highly basic region which has been reported to be essential for lytic activity.
The structure of Melittin, 1982
Melittin exemplifies this large class of membrane-active peptides that manifest membrane-disrupting activity when incorporated into traditional bilayer delivery systems (i.e., liposomes).
It is a very nonspecific cytolytic peptide that attacks all lipid membranes, leading to significant toxicity when injected i.v.; the basis of its action is a physical and chemical disruption of membrane structure, resulting in profound compromise of the cell permeability barrier.
Melittin forms ion channels which span membrane bilayers through the tetrameric association of melittin monomers; the channels are more permeable to anions than to cations, probably due to the accumulation of positive charges on the C-terminal region.

Melittin forms channels in lipid bilayers, 1981
Depending on the orientation of Melittin, pores are formed.
The orientation of Melittin in membranes is dependent on the lipid composition and the physical condition of the membrane; one of the key parameters that determinate the orientation of membrane-bound peptides, is their concentration.
At low concentration, Melittin adopts parallel orientation with respect of the membrane surface, whereas when the concentration increase above a certain threshold value, an increasing fraction of peptide molecules change to the perpendicular orientation.
Melittin induced trans membrane pores are present only when the peptide is orientated perpendicular to the membrane bilayer.
Orientation and Dynamics of Melittin, 2007
about orientation
The residue Lys 7, located in the hydrophobic moiety of the helix, and residues Lys 23 , Arg 24, Gln 25, and Gln 26 at the C-terminus hydrophilic, form hydrogen bonds with water molecules and with the ester carbonyl groups of the lipids. Lipid acyl chains are closely packed around melittin, contributing to the stable association with the membrane. Melittin has effects on both layers of the membrane: the presence of melittin in the upper layer, causes a local thinning of the bilayer that favors the penetration of water. As Cl− ions entered into the prepore, they are electrostatically entrapped by positively charged residues located at its wall; reorientation of the local lipids occurs as their flipping.
Melittin forms a short-lived pore; upon the disintegration of the pore, a fraction of the peptides translocates across the lipid bilayer.
Pore formation and traslocation of Melittin, 1997
The aim of this study is to achieve a practical therapeutic application by designing a synthetic delivery vehicle that would serve as a protected peptide storage platform, sufficiently stable to deliver the melittin, by either “passive” accumulation, using endothelial permeability and retention ( EPR ), or “active” delivery by selective molecular recognition to either advanced or preclinical tumors.
EPR is caused by the greater leakiness of blood vessels in the vicinity of the tumor, which allows substances, such as nanoparticles, to escape more readily from the bloodstream into nearby tissues, and the generally poor drainage of lymph from tumors, which helps trap substances in tumor tissue.
So far, the in vivo application of cytolytic peptides for cancer therapeutics is hampered by toxicity, nonspecificity, and degradation.
Even if cytolytic peptides are an attractive class of anticancer candidates because of their wide‐spectrum lytic properties, their therapeutic potential cannot be realized without a proper delivery vehicle, because of their off‐target toxicity, nonspecificity, and unfavorable pharmacokinetics.
Pharmacological requirements for such a delivery vehicle, would include suitable pharmacokinetic properties, indicative of avoidance of rapid destruction by proteolysis, cell-selective targeting and lack of significant off-target toxicity.
Process challenges would include the need to preserve the complex peptide/protein structure throughout the formulation process, which is required to retain biological activity.
Based on these needs, perfluorocarbon nanoemulsion vesicle has been used (approximately 227nm).
The nonspecific amphipathic cytolytic peptide melittin, is incorporated into the outer lipid monolayer of a perfluorocarbon nanoparticle; this particle consist of a hydrophobic PFC core, surrounded by a lipid surfactant monolayer; PFC is clear, colorless, odorless, nonconducting, and nonflammable, very chemically stable, and is not metabolized in body tissues.
These ‘nanobees’ appear capable of serving as a selectively targeted carrier, that remains stable after insertion of melittin.

National Cancer Institute
Samuel Wickline, M.D., head of the Siteman Center of Cancer Nanotechnology Excellence at Washington University, says:
"The nanobees fly in, land on the surface of cells and deposit their cargo of melittin which rapidly merges with the target cells. […] Melittin is a workhorse, it's very stable on the nanoparticles, and it's easily and cheaply produced. We can add melittin to our nanoparticles after they are built […] Melittin fortunately goes onto the nanoparticles very quickly and completely and remains on the nanobee until cell contact is made.”
The study demonstrate the ability to safely deliver significant payloads of melittin i.v. and to target and kill three types of experimental tumors:
- Syngeneic (genetically identical) (B16F10 mouse melanoma);
- Xenograft (a tissue graft obtained from a donor of a different species from the recipient) (MDA-MB-435 human breast cancer);
- Precancerous lesions in K14-HPV16 mice with squamous dysplasia and carcinoma.
Furthermore, it is illustrated the flexibility of this approach in both actively and passively targeted applications in vivo, characterize the unique fusion-based delivery mechanism of these nanovehicles.
RESULTS
Melittin-loaded nanoparticle synthesis and action on cancer cells in vitro
When melittin is incorporated into the nanoparticle’s stabilizing outer lipid monolayer, it produces no detectable alteration in the basic nanoparticle structure. In contrast, melittin completely destroys liposomal carriers by membrane disruption through pore formation.
Fig.1
To confirm the protection against off-target cell toxicity afforded by the melittin-loaded nanoparticles, red blood cell hemolysis assays were conducted in vitro. Soluble free melittin rapidly lyses red cells, which is a major source of in vivo toxicity and serves as a standard test of melittin’s bioeffects.
Incorporation of melittin on the nanoparticles, increased the IC50 (concentration that causes 50% hemolysis) from 0.51 ± 0.12 μM for free melittin to greater than 10 μM for nanoemulsions (fig. 1B). Even a concentration of 25 μM nanoparticle melittin only elicited 10% hemolysis.
When tested in vitro as a cancer cell therapeutic, free melittin was highly toxic to B16F10 melanoma cells (IC50: 0.75 ± 0.19 μM); however, when incorporated on the nanoparticle, the IC50 was 5.1 ± 1.2 μM, which reflects the slow release of melittin from the nanoparticles.
Melittin-loaded nanoparticle safety in vivo
Table 1
As we can see, no significant changes in the levels of serum enzymes or electrolytes were observed that might otherwise suggest damage to major organs after injection of melittin-loaded nanoparticles (mice treated with saline are used as a valid internal control).
Also, routine tissue histology on liver, lung, kidney, and heart revealed no evidence of tissue damage; tests for free hemoglobin in the urine with urine strips revealed no evidence of hemolysis.
Melittin-loaded nanoparticle suppression of tumor growth in vivo by EPR
The accumulation of drug-delivery vehicles in solid tumors by the endothelial permeability and retention effect ( EPR ), is well documented and is the dominant mechanism of action for the therapeutic effect of the FDA-approved compounds Doxil and Abraxane (nanoparticle cancer treatement). It has been demonstrate that this approach is simple and acceptable as a form of therapy.
The study tested the efficacy of melittin-loaded nanoparticles against xenograft MDA-MB-435 human breast cancers in athymic nude mice.
The melittin-loaded nanoparticles significantly inhibited the growth of the tumors by 24.68% ± 1.57% as compared with the saline controls.
Given the prominent role played by the immune system in effecting tumor regression, we also evaluated the efficacy of melittin-loaded nanoparticles in immunocompetent mice. The final tumor weight on day 14 was 0.23 ± 0.19 g for the treated mice, compared with 1.87 ± 0.86 g for the saline controls (or ~88% reduction in tumor mass). Histology revealed a decrease in the number of blood vessels (CD31), a decrease in proliferating cells, and significant areas of necrosis (H&E) in the melittin-loaded nanoparticle–treated tumors, that were not present in saline controls.
Fig.3
Regarding the safety profile in a cancer model, no toxic effects were apparent in terms of the organ weights or serum chemistry profiles.
Development and characterization of molecularly guided nanoparticles
Although nanoparticles are effective as cargo vehicles to deliver the melittin to established tumors by EPR, the study want to determine whether specific molecular targeting of the nanoparticles to neovascular targets, would be useful in treating nascent early-stage tumors or precancerous lesions that are just entering an aggressive angiogenic phase.
Human melanoma (C32) cells that express the αvβ3 integrin were used in vitro to assess the interactions of targeted nanoparticles with cell membranes.
Nanoparticles were targeted by incorporating a peptidomimetic αvβ3 integrin–binding ligand and the proliferation of mouse endothelial (2F2B) cells and human melanoma (C32) cells in culture was tested.
By including αvβ3 integrin mimetic on the nanoparticles, the interaction of nanoparticles with cells was increased 4-fold. This increase, resulted in a reduction of the IC50 for inhibition of cell proliferation to 6.8 ± 2.14 μM for C32 cells and 8.39 ± 2.35 for 2F2B cells.
Thus, the increased binding of the nanoparticles to the cells accounts for the increased activity of the targeted nanoparticles.
Since the nanoparticles themselves do not cause any deleterious effects on the cells, by using them as nanocarriers, we attenuated nonspecific cellular lysis that is characteristic of soluble (free) melittin and simultaneously enabled selective targeting of the nanoparticles to endothelial and cancer cells and restored the lytic potential of melittin in proportion to the extent of cell-nanoparticle interaction. The activity of the targeted melittin-loaded nanoparticle complex, closely approximates the lytic activity of free melittin but now is restricted to the cancer and endothelial cells that express the integrin.
Fig.4
In the absence of the targeting peptidomimetic, very few nanoparticles were associated with the surface membrane of the cells (Figure 4C). However, when the targeted nanoparticles were presented to the cells, large numbers were associated with microvilli and also extended to the membrane surface (Figure 4D).
Cell-surface contact and nanoparticle-cell membrane interactions offer a direct pathway for cargo transport from the nanoparticle: the amphiphile stabilizing layer allows the formation of a hemifusion pathway between the lipid monolayer of the nanoparticle and the bilayer of the cell, that leads to the delivery of peptide cargo driven by a combination of surface tension and chemical potential.
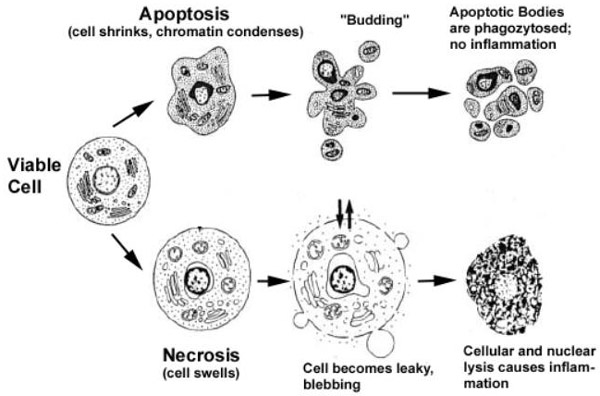
Targeted melittin-loaded nanoparticles induce cell death by apoptosis
To further elucidate the targeted delivery mechanism, we labeled melittin with fluorescein and examined the trafficking of the peptide (1 μM) delivered from nanoparticles to cells.
Fig.5
With this analysis, it is shown the contribution of apoptotic versus necrotic cellular death, after the interaction with targeted melittin-loaded nanoparticles.
A dose-dependent decrease was observed in the percentage of viable cells after incubation with αvβ3 integrin–targeted melittin-loaded nanoparticles.
The cell death was explained by the observed dose-dependent increase in early and late apoptotic cells (Figure 5C).
To determine whether the nanoparticle-delivered melittin provoke apoptosis, after trafficking to intracellular membranes via activation of the intrinsic pathway, the intracellular release of cytochrome c has been measured; to delineate the role of intracellular membrane disruption versus cell-surface membrane lysis, we also determined the release of lactate dehydrogenase (LDH) from cultured C32 melanoma cells.


The cells treated with targeted melittin-loaded nanoparticles released cytochrome c from their mitochondria and exhibited a concentration-dependent increase of apoptosis (Figure 5D). However, there were small releases of LDH after treatment with 2.5 μM (11% ± 5%) or 12.5 μM (17% ± 3%) αvβ3 integrin–targeted melittin-loaded nanoparticles; the corresponding release of cytochrome c was 23.5% ± 4.3% and 37.2% ± 6.1%, respectively, which was significantly higher than in the control cells. Thus, the cellular release of LDH did not increase to the same extent as the release of cytochrome c, while the fraction of apoptotic cells progressively increased.
In contrast, the free melittin doses that produced necrotic cell death generated the expected concomitant release of both cytochrome c and LDH.
These data demonstrate a clear distinction between the effects of melittin delivered to the cell surface by nanoparticles, and the activity of soluble (free) melittin. Even at high concentrations, melittin delivered to the cell surface from nanoparticles drives cellular apoptosis and does not produce the necrotic cell death that is induced by soluble melittin. These data suggest that melittin is being internalized by an active process after the cell-surface delivery by the nanoparticles. Yet considerable melittin also remained on the cell surface, where its pore-forming activity was held in check by some mechanism.
Role of cholesterol in the induction of apoptotic cell death by αvβ3 integrin–targeted melittin-loaded nanoparticles
Cholesterol is an amphipathic molecule that helps maintain the integrity and fluidity of membranes.
Lipid rafts/caveolae are membrane platforms for signaling molecules that regulate various cellular functions, including cell survival; the are low-density, detergent-resistant microdomains of plasma membrane that are enriched in cholesterol and glycosphingolipids. Because cholesterol is an essential lipid component of rafts/caveolae implicated in Akt activation, it is accumulated in several tumors; this because Akt/protein kinase B (PKB) is a serine/threonine kinase that is a critical regulator for cell survival and proliferation, especially in human malignant cancer. Activated Akt, phosphorylates pro-apoptotic proteins, inactivating their activities while also up-regulates anti-apoptotic genes such as Bcl-xL and FLICE-inhibitory protein (FLIP), driving to tumor cells survival.
Studies, have demonstrate that cholesterol depletion activates death-related pathways, leading to the anoikis-like apoptosis of various cancer cell types, along with down-regulation of Akt activity and Bcl-xL and caspase-3 activation; on the other hand, reconstitution of rafts/caveolae by cholesterol addition, reactivates Akt and restored cell viability.
Colesterol-rich lipidic rafts in cancer cells, 2006
Melittin-Lipid Bilayer Interactions and the Role of Cholesterol, 2008
The presence of cholesterol in the plasma membrane of C32 cells, might inhibit local pore formation, which would in turn reduce cell necrosis.
Because cancer cells may hide greater levels of membrane cholesterol than do normal cells, in order to reduce apoptosis, it was thought that depletion of cholesterol might alter the mode of cell death.
Fig.6
To evaluate this hypothesis, cholesterol was depleted from the cell plasma membrane, to identify the cause of cellular mortality in response to targeted melittin-loaded nanoparticles. Modest reductions of cell membrane cholesterol (7%-12%) produced no untoward effects on cell proliferation; however, the cholesterol-depleted cells displayed a decrease in proliferation when exposed to targeted melittin-loaded nanoparticles (fig.6A).
The removal of approximately 13% of cellular cholesterol before treatment with 2.5 μM melittin on αvβ3integrin–targeted nanoparticles, generate inanimate cell debris (fragments of devitalized tissue or foreign matter), consistent with a completely necrotic mode of cell death.
As shown in Fig.6H, the nanoparticles were stabilized by a monolayer of lecithin and contained adsorbed melittin; the αvβ3 peptidomimetic was bound to the integrin on the cell membrane surface; a bilayer normally containing 40% cholesterol.
The close approximation of these 2 lipid layers leads to the formation of hemifusion stalk; this provides a pathway for the movement of lecithin and melittin to move to the cell surface.
In the high cholesterol membrane, melittin is unable to form pores but through endocytosis moves to the intracellular membranes like late endosomes, endoplasmic reticulum, and mitochondrial outer membrane, all of which have low cholesterol content; here Melittin is able to form pores, leading to apoptotic cell death.
When cholesterol is reduced at the cell surface, melittin-induced pores compromise the osmotic control of the cell, leading to necrosis.
Safe regression of precancerous lesions in vivo with targeted melittin-loaded nanoparticles
The K14-HPV16 mouse model is an example of a genetically engineered model of squamous carcinoma harboring human papilloma virus (HPV) transgenic elements (E6 and E7 oncogenes), driven by a keratin promoter. These mice progress from hyperkeratosis (1 month) to acanthosis and papillomatosis (4 to 5 months) and finally to epidermal carcinoma (7 to 12 months).
Angiogenesis is abundantly observed near the basement membrane, between the dermis and epidermis, during the precancerous dysplastic stage and persists in invasive squamous cancers. Overexpression of integrin αvβ3 receptors on endothelial cells is a hallmark of angiogenesis.
Fig.7
The therapeutic effect of αvβ3 integrin–targeted melittin-loaded nanoparticles has been evaluated, after the hypothesis of its a dual antiangiogenic and antitumor action on epidermal papillae. The efficacy of the targeted therapeutic in vivo was demonstrated by a quantitative reduction in dermal papillae, which are the site of the genetically driven dysplastic changes. The mice treated with αvβ3 integrin–targeted melittin-loaded nanoparticles, comparing to the onesaline-trated, shows an 80% reduction in severe dysplastic lesion prevalence.
No apparent organ toxicity was observed after all treatments, as indicated by unchanged blood chemistry profiles.
DISCUSSION
In designing this delivery vehicle for melittin, the aim was to limit the off-target membrane lytic activity of this peptide, without having to alter its native structure by mutagenesis or fusion.
In contrast with melittin delivered from nanoparticles, free melittin rapidly inserts into the red cell membranes after i.v. administration. At high doses, this causes massive hemolysis and acute renal failure and at low doses, subtle changes in red cell rigidity. In any case, the rigid, deformed, or lysed red blood cells are rapidly eliminated from the circulation by the spleen, liver, and lungs.
The absence of any apparent toxicity of melittin-loaded nanoparticles after injection of an equivalent dose of free melittin, points to the effective shield that the nanoparticle lipid monolayer provides against the cytotoxic effects of melittin.
Incorporation of melittin onto the nanoparticles, prolongs the melittin circulation time, thereby offering an increased stochastic probability of accumulation in the tumor and targeted binding to sites of angiogenesis.
The random collisions that nanoparticles encounter with different cell types in the vasculature (i.e., red and white blood cells, endothelial cells) might trigger the formation of a hemifusion complex and subsequent delivery of small amounts of melittin to the cells during their prolonged serum residence times compared with free melittin; however, this delivery is small and produces a striking decrease in nonspecific hemolysis attributable to the use of nanoparticles as compared with free melittin.
The demonstrated efficacy of both nontargeted and αvβ3 integrin–targeted melittin-loaded nanoparticles, indicates that the melittin-loaded nanoparticles could exert their antitumor effect either by nonspecific trapping in the abnormal tumor vasculature or binding to overexpressed integrins on angiogenic endothelial cells.
In each case that we examined, efficacy is clearly demonstrable regardless of immune state, tumor type, animal background, or mechanism of nanoparticle interaction with the tumor (EPR vs. targeted delivery).
The interaction of molecularly targeted nanoparticles with cells, differs dramatically from that of other typical biovehicles (e.g., liposomes, polymeric nanoparticles), in that it does not seem to require endocytosis of the intact carrier complex.
Specific targeting to cancer cells by the incorporation of mimetic targeting ligands, leads to the binding, close apposition, and hemifusion of the lipid monolayer with the targeted cell lipid membrane.
Lipids and peptides, such as melittin, that are inserted in the nanoparticle monolayer can then diffuse along the continuous hemifusion pore, becoming part of the target cell membrane.
Entry into the cell appears to be by endocytosis of the cellular membrane to which the cargo has been transferred.
This study demonstrate that the contact-mediated transfer of melittin by nanoparticles, produces cytochrome c release and apoptosis. Previously, cellular apoptosis from melittin exposure of cells has been attributed to calcium entry or activation of phospholipase A2.
The details of the nanoparticle mechanism of melittin delivery remain to be revealed, but the exclusive apoptotic cell destruction suggests that the cell-surface membrane remains intact and that intracellular membranes are being disrupted.