Definition
We speak of hypertrichosis commonly to refer to any situation in which there is an objective, or even subjective, excess of hair.
Since hair growth is abundant and typical in males, hypertrichosis, by definition, refers exclusively to the female sex.
In the strict sense is defined hypertrichosis the excess development of hair in regions where these are normally present in the woman. The term hirsutism indicates the appearance of hairiness in women in areas normally hairless, then when this hairiness is remarkable, remembering the hair of men, it is called virilism follicle.

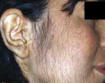
Epidemiology
The development of the follicle is influenced by genetic factors-racial. The incidence of hypertrichosis is insignificant in the yellow race and the black one, is less common in Northern Europe, while it is very common in people who originate from the Mediterranean area.
Some syndromes such as congenital adrenal hyperplasia are genetically transmitted. The idiopathic hirsutism, is often a family picture. It appears at menarche, and increasing is slowly progressive.
Diagnosis and Symptoms
The diagnosis of hirsutism is largely culturally determined: this condition is acceptable in some societies and unacceptable in others (where women see in a hypertrichosis, however small, a serious loss of femininity). The hypertrichosis is rarely caused by serious diseases, in most cases is the clinical manifestation of functional disorders of different type; also the same type of disorder can cause a great individual variability in terms of clinical distribution of hairs and clinical symptoms associated. Even hormonal parameters can change spontaneously during the clinical follow-up of patients in relation to changes in secretion, metabolism and mechanism of action of androgens. Therefore, even if the etiological classification of hypertrichosis may seem simple, defining a patient "hypertrichotic" on the basis of biochemical parameters at our disposal is often impossible.
The objective assessment is carried out with the Ferriman-Gallwey score. It assigns a score from 0 (no hair) to 4 (clearly virile presence of hairs), for each of the nine areas of the skin settings. Remember how often the signs of androgenic activity are disguised or removed from female patients.
In performing the physical examination should also search:
- Signs of Cushing. The presence of striae rubrae, lunaris facies, buffalo hump, etc.. push to investigate adrenal function.
- Signs of acromegaly. The facies and other alterations present in acromegalic patients, allow diagnosis with observation alone.
- Obesity. Possible presence of polycystic ovaries.
- Acanthosis nigricans. Hyperkeratosis papillomatosa with deposition of melanin, located at the posterior region of the neck, axilla, groin and the general in points of friction.
- Blood pressure. We have an increase in blood pressure in patients with inadequate 11-hydroxylase in Cushing, acromegaly.
Patogenesis of Hypertrichosis hormone-mediated
Androgens are capable of stimulating the growth of sexual hair. In women, the sources of androgens are several and varied. Testosterone (T) comes mainly from the peripheral conversion of other steroid precursors (androstenedione in particular) and to a lesser extent by the adrenal and ovarian secretion. Dihydrotestosterone (DHT) comes entirely from the peripheral conversion of androstenedione and testosterone as its glandular secretion irrelevant under physiological conditions. Androstenedione, dehydroepiandrosterone (DHEA) and androstenediol come instead largely by ovarian and adrenal secretion and to a lesser extent from the processing device of precursors. Dehydroepiandrosterone sulfate comes only from the adrenal secretion (DHEAS).
The origin of certain androgens and their circulating levels change as a function of the menstrual period (ovarian secretion of androgens is maximum in the late follicular phase). For this reason, the best time for sampling is the 2nd day of the cycle. All androgens are secreted episodically and many of them possess a circadian rhythm. In the blood, certain androgens (T and DHT for example) circulate bound to a specific globulin (which has a high affinity but low binding capacity) called sex hormone binding globulin (SHBG), other androgens instead are more loosely bound to albumin (a high capacity and low affinity ).
One of the key aspects of the mechanism of action of androgens in Hirsutism is their metabolism at the level of target structures. Only a small part of the circulating DHT originates from tissular metabolism of testosterone, while the majority comes from hepatic conversion. The same is true for the 5alpha-androstan-3alpha-17beta-diol, a metabolite of DHT.
From this it appears that dihydrotestosterone and 3alpha-diol circulating can not be in any way a humoral indicator of the metabolic activity of the androgen target tissues. On the contrary the 3alpha-diol glucuronide circulating comes largely from the conversion of DHT, more precisely 50% in males and almost 100% in females.
Consequently the 3alpha-diol glucuronide can be considered the more specific biochemical marker of androgenic activity at the level of the targets in the woman and the biochemical marker of hirsutism. Find high blood levels of 3alpha-diol glucuronide in women is equivalent to a diagnosis of hypertrichosis or hirsutism hormone-mediated.
On the other hand, since this androgen is the expression of androgenic hypermetabolism in the skin, the findings of elevated levels of 3alpha-diol glucuronide does not pose any etiologic diagnosis, it is equivalent only to the clinical finding of the presence of the increase in hair but indicates that this is mediated by hormonal activity device.
The determination of androgens in the blood is of little relevance to the differential diagnosis of polycystic ovary syndrome or idiopathic hirsutism.
Except for cancer, congenital adrenal hyperplasia and polycystic ovaries is often very difficult to determine the origin of hyperandrogenism in the course of hypertrichosis or hirsutism.
All disorders associated with hirsutism or virilism of the adrenal cortex are accompanied by an increase of 17 ketosteroidi note that these compounds are so called because they have a keto group in position 17 and are neutral, therefore the testosterone is not included between them.
The history will often be much guidance for the pathogenesis of hirsutism.
A hypertrichosis in a woman of reproductive age with regular menstrual cycle is unlikely to be supported by a hormonal disorder.
Heavy menstruation or menorrhagia will suggest to anovulatory cycles.
The presence of premenstrual syndrome will suspect a failure progestin.
A hirsutism with notes of masculinization, incipient androgenetic outflow and amenorrhea will suspect a congenital adrenal deficiency.
A hirsutism with distinct notes of virilism risen rapidly in women in the third to fourth decade of life will be guidance for a neoplastic disease.

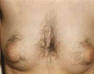
Major hormonal causes of hypertrichosis and hirsutism
- Idiopathic sensivity increased to androgens;
- Subtle ovarian or adrenal alterations;
- Ovarian diseases -> PolyCystic Ovary Syndrome;
- Androgen-producing tumors;
- Adrenal diseases -> Cushing's syndrome;
- Congenital adrenal Hyperplasia;
- Glucocorticoids;
- Endocrinopathies -> hyperprolactinemia;
- Acromegaly;
- Hypo-Hyperthyroidism;
- Obesity;
- Hirsutism due to medication.
In the case of hypertrichosis, hirsutism or hormonal virilism we propose the following basic tests to be carried out in early follicolinic phase of the cycle (by day 7) when all the hormones involved have values close to the baseline:
3 al-diol-g, FSH - LH, cortisol, androstenedione, 17 OH progesterone, testosterone, prolactin, 17 beta estradiol, progesterone.
In the luteal phase will then repeated the dosage of progesterone.
At any time of the cycle will be possible to measure DHEA-S, cortisol, TSH and fT4.
Moreover, the hair follicles were able to interact with androgens as they possess the receptors, and in relation to their sensitivity to androgens are three different types of hair: sexual, non-sexual, ambisexual. Therefore Hypertrichosis and Idiopathic Hirsutism may also be due to an increased sensitivity of skin receptors to androgens secreted even in normal quantities, or to other alterations of the receptors.
(Hirsutism causes. 2010)
Hormones Involved
3 alpha-diol glucuronide (3 al-diol-g)
High Serum values of 3 al-diol-g make unequivocally differential diagnosis of hirsutism and hypertrichosis mediated hormonal activity is not mediated by hormonal activity.
Unfortunately, at present, this dosage can not be done routinely but only as a sophisticated survey research and, as far as we know, there aren't laboratories able to dose it fluently.
(3 alpha, 17 beta-androstanediol glucuronide in plasma. A marker of androgen action in idiopathic hirsutism. 1982)

Gonadotropins (FSH - LH)
The values of normal plasmtic FSH and LH in women in the follicular phase range from 5 to 25 U / L. The ratio between the two gonadotropins at this stage is normally of 2/1.
Elevated plasma levels of gonadotropin luteinizing hormone (LH) and gonadotropin follicle stimulating hormone (FSH) normal or decreased, together with high concentrations of androstenedione, oriented towards the polycystic ovary syndrome.
Dehydroepiandrosterone sulfate (DHEA-S)
Dehydroepiandrosterone sulfate is a weak androgen secreted almost exclusively by the adrenal gland (99%). Circulates in the blood bound to albumin.
In women, the values of normal plasmatic DHEA-S range from 2.1 to 8.8 mM / L and decrease after menopause falling well below 0.3 -1.6 mmol / L.
Plasma levels ranging from 15 to 30 mM / L point to a diagnosis of normal adrenal hyperandrogenism, if associated with hypercortisolemia point to a Cushing's syndrome if over 50 to 100 mM / L to a malignant disease, if finally associated with increased rates 17-OH progesterone and ipocortisolemia to a congenital enzyme deficiency.
(Dehydroepiandrosterone Sulfate (DHEA-S), Serum. 2008)
Delta 4 androstenedione (D4)
This hormone is the most 'important androgen in women and is largely responsible for the female of acne and hirsutism and androgenetic outflow.
Comes to 50% of the ovarian secretion, by 30% from the adrenal secretion and for the remaining part of the peripheral conversion of precursors. The ovary secretes androstenedione particularly during the interim period and luteal cycle. The production of androstenedione may increase premenopausal resulting in mild virilization.
Androstenedione forms testosterone by the enzyme 17 beta hydroxysteroid oxidoreductase. Androstenedione and testosterone represent intermediate stages of estrogen biosynthesis.
The normal values of androstenedione in women are usually of between 1.33 and 5.27 nmol / L.
Values ranging between 10 and 20 nmol / L, associated with high gonadotropin luteinizing hormone (LH), orient towards a possible diagnosis of polycystic ovaries. Values above 30 nmol / L are orientative instead for an adrenal or ovarian hormone-secreting tumor.
17 OH progesterone (17 OH D)
The increase of this hormone in plasma levels to above 15 nmol / L in women in the follicular phase, especially if these high rates are accompanied by high values of DHEA-S and low or normal values of cortisol, orients toward the diagnosis of adrenal enzyme deficiency 21 or 11-hydroxylase.
Testosterone (T)
Normal values in women range from 0.02 to 0.8 ng / ml (0.3 - 3 nmol / L).
Testosterone in plasma is bound to 99%, approximately, to one specific betaglobulina: Sex Hormone Binding Globulin (SHBG). Only the free part is metabolically active.
Since the binding capacity of SHBG for testosterone is around 14 ng / ml, so well above the serum testosterone level, the free hormone dosage is practically useless and this will be always constant fraction of total hormone.
(Testosterone and Androstenedione Blood Production Rates in Normal Women and Women with Idiopathic Hirsutism or Polycystic Ovaries. 1967)


Prolactin (PRL)
Normal values in women are around 9-20 ng / ml or 75 to 504 mU / L during pregnancy and rise to 150 to 200 ng / ml or 3000-4000 mU / L.
In the course of hyperprolactinemia, whatever its origin, it is frequent a hypertrichosis and a outflow in telogen, perhaps for activation by prolactin of 5 alpha reductase. Hyperprolactinemia can be induced (by estrogen, by psycholeptics etc.), Functional (from stress, lactation etc), from pituitary adenoma. The probability of a pituitary adenoma is directly correlated with the values of hyperprolactinemia. Prolactin values > 200 ng / ml or 5,000 mU / L, outside of pregnancy and lactation, are almost invariably associated with the presence of a pituitary adenoma and values between 100 and 200 ng / ml or 2000-5000 mU / L is believed to be very suspicious. A hyperprolactinemia anamnestically not always clearly requires further diagnostic tests.
(Prolactin in hirsute women: possible roles for androgens in suppressing basal levels, and for estrogens in enhancing TRH-induced responses. 1984)
17 beta estradiol
In early follicolinic phase 17-beta-E woman values will be between 35 - 100 ng / L or 60 to 200 pmol / L.
Progesterone
In early follicolinic phase levels of progesterone, will be basal, comparable to those of the male and including between 0.2 to 0.9 ng / ml or <2 nM / L but in luteal phase will rise above 8 ng / ml or> 25 nM / L, proving the ovulation. A failure to increase the progesteronemia to 22 ° - 24 ° day of the cycle will make the diagnosis of anovulation.
Cortisol
High concentrations, especially in the evening, orient for Cushing's syndrome. In Cushing's syndrome the baseline cortisol is increased and the circadian rhythm disappeared.
The normal values at 8 am are between 0.14 to 0.63 mM / L (160-690 nmol / L), at 16 between 0.083 to 0.42 mM / L (50 -250 nmol / L), the 20 hours is less than 50% of the value 8 o'clock.
Low or normal cortisol values associated with increased concentrations of dehydroepiandrosterone (DHEA) and 17-OH progesterone (17 OH D) are pathognomonic of adrenal enzyme deficiency (21 or 11 hydroxylase) syndrome with virilization.
Thyroid hormones (fT4, TSH)
Hypothyroidism is frequently accompanied by hypertrichosis.
It can make a diagnosis of hypothyroidism by dosing the thyroid stimulating hormone (TSH) and thyroid hormones, in particular thyroxine (T4). A primary hypothyroidism is characterized by high plasma levels of TSH and low or normal T4.
The values of normal plasma T4 are between 5 and 12 mg / dl or 65 nanomoles -155 / L; normal values of T3 are between 115 and 190 ng / dl or 3.54 to 10.16 picomoles / L; the normal values of TSH are between 0.5 and 4 mU / L. As hypothyroidism is passed on the apparatus follicle is not clear, however we know that the TSH releasing factor pituitary prolactin and hypothyroidism (even latent) is often accompanied with hyperprolactinemia, thyroid hormones also modulate the plasma concentration of the protein carrier of testosterone and estrogen (SHBG) and decreases in this case also latent hypothyroidism (high TSH and low or normal values of T4), causing also an skin androgenism to increase plasma free testosterone. Also thyroxine is one of the main stimulus for the production of epidermal growth factor (EGF), which certainly in some animals, and perhaps also in humans, modulates the growth of hair.
(Thyroid Hormone Disorders. 2001)

Therapy
The decision on when to start therapy and the type of treatment of hirsutism should be taken based on the index of Ferriman and Gallwey.
The therapy also be adapted to the needs of the patient Tenedos realize that treatment is often long-term (at least 6-12 months) for the slowness with which the therapeutic effects appear and that the underlying cause is not definitively resolved by therapy that is symptomatic and therefore hirsutism tends to recur after discontinuation of therapy.
Idiopathic Hirsutism
In idiopathic Hirsutism due to increased peripheral sensitivity to androgens, find their full scope the receptor antiandrogens, cyproterone acetate and spironolactone, which act by inhibiting the binding of testosterone to DHT. These should always be associated with contraceptive therapy to avoid, in the case of pregnancy, abnormal sexual development of a male fetus.
(Hirsutism Idiopathic. 2010)
- a) Cyproterone acetate (Androcur) 50 - 100 mg per day associated with the first 10 days of a cycle estroprogestin (usually the Diane).
- b) Spironolactone (Aldactone) 25 - 100 mg associated with the last 10 - 16 days cycle of an estrogen-progestagen (usually Diane).
Androcur, Diane, Aldactone can all be associated together according to a cyclical pattern that includes 10 days of Androcur + Diane, Diane one day only, 10 days Aldactone 100 + Diane, seven days a pause and resume the cycle.
- c) Flutamide (Eulexin capsules) 250 - 500 mg per day without interruption associated with one estroprogestin cyclic.
Positive results, but sometimes only temporary, after you have 6-12 months of these therapies. Should always be reminded of the general contraindications to these therapies: pregnancy, age > 35 years if the woman is smoke or hypertension, previous thromboembolism, liver failure, suspected estrogen-dependent neoplasia.



Cyproterone acetate ________________ Spironolactone ____________________ Flutamide
In Hyperandrogenism mainly ovarian (polycystic ovary syndrome, or PCOS)
Progestogens, common oral contraceptives:
- a) Containing progestins with low androgenic activity such as linesterolo (Lyndiol, Franovul, Minifal) or desogestrel (Planum, Practil 21).
- b) Progestogens containing cyproterone as progestin progestogens: 0.035 mcg ethinyl estradiol + 2 mg cyproterone acetate (Diane).
(Endocrinology of Hirsutism. 2010)
In defects of adrenal steroidogenesis:
Mainly of 21-hydroxylase deficiency or 11-beta hydroxylase, in the form "late", are used glucocorticoid dexamethasone 5 mg or 0.25 to 0.5 mg at 23.
In addition to suppress the secretion of ACTH, and then the synthesis of adrenal androgens, glucocorticoids reduce, in part, also ovarian steroidogenesis.
The side effects are minimized by the administration of low doses.
If the plasma levels of DHEA-S are removed, you can expect to get a therapeutic success in 50% of cases after 4-6 months.
(Defects of adrenal steroidogenesis in patients with hirsutism. 1998)
Also:
In case of hyperprolactinemia and acromegaly surgical removal of the adenoma or bromocriptine.
In case of dysthyroidism this must be corrected.
In case of ovarian or adrenal removal of the same is often curative also for the hypertrichosis.
In obesity, the only weight reduction can lead to an improvement of hirsutism.