Avetta Cecilia
Devecchi Andrea
INTRODUCTION
Parkinson's disease(PD also known as idiopathic or primary parkinsonism, hypokinetic rigid syndrome/HRS, or paralysis agitans) is a degenerative disorder of the central nervous system. The motor symptoms of Parkinson's disease result from the death of dopamine-generating cells in the substantia nigra, a region of the midbrain; the cause of this cell death is unknown. Early in the course of the disease, the most obvious symptoms are movement-related; these include shaking, rigidity, slowness of movement and difficulty with walking and gait. Later, thinking and behavioral problems may arise, with dementia commonly occurring in the advanced stages of the disease, whereas depression is the most common psychiatric symptom. Other symptoms include sensory, sleep and emotional problems. Parkinson's disease is more common in older people, with most cases occurring after the age of 50. Parkinson’s disease (PD) is characterized by reduced flexibility, conceptualization, and visuo-spatial abilities. Although these are essential to creativity, case studies show emergence of creativity during PD. Knowledge about the role of dopamine in creativity so far only stems from a few case reports.
Eugénie Lhommée,1,2,3,* Alina Batir,1,2,3 Jean-Louis Quesada,4 Claire Ardouin,1,2,3 Valérie Fraix,1,2,3 Eric Seigneuret,1,2,5 Stéphan Chabardès,1,2,5 Alim-Louis Benabid,5,† Pierre Pollak,1,2,3,† and Paul Krack1,2,3 (2014). Dopamine and the Biology of Creativity: Lessons from Parkinson’s Disease
PHYSIOLOGY
Dopaminergic neurons (i.e., neurons whose primary neurotransmitter is dopamine) are comparatively few in number — a total of around 400,000 in the human brain— and their cell bodies are confined to a few relatively small brain areas, but they send projections to many other brain areas and exert powerful effects on their targets. These dopaminergic cell groups were first mapped in 1964 by Annica Dahlström and Kjell Fuxe, who assigned them labels starting with the letter "A" (for "aminergic"). In their scheme, areas A1 through A7 contain the neurotransmitter norepinephrine, whereas A8 through A14 contain dopamine. Here is a list of the dopaminergic areas they identified:
The substantia nigra, a small midbrain area that forms a component of the basal ganglia. The dopamine neurons are found mainly in a part of this structure called the pars compacta (cell group A8) and nearby (group A9). In rodents, their most important projections go to the striatum, globus pallidus, and subthalamic nucleus, all of which also belong to the basal ganglia, and play important roles in motor control. The name substantia nigra is Latin for "dark substance", and refers to the fact that the dopaminergic neurons there are darkly pigmented. These neurons are especially vulnerable to damage. When a large fraction of them die, the result is a Parkinsonian syndrome.
The ventral tegmental area (VTA), another midbrain area. This cell group (A10) is the largest group of dopaminergic cells in the human brain, though still quite small in absolute terms. Projections from these dopaminergic neurons go to the nucleus accumbens and the prefrontal cortex as well as several other areas.These neurons play a central role in reward and other aspects of motivation. The nucleus accumbens is often considered to be the "limbic" part of the striatum. As such, it is the part of the striatum involved in the highest level aspects of motor control, which include motivation and decision-making. Thus, the role of the VTA in motivation and decision-making is structurally analogous to the role of the substantia nigra in low-level motor control. In primates (i.e. monkeys and humans), the dopamine neurons from the regions of the substantia nigra and VTA project throughout most of the cortical mantle, with particularly dense innervation of the motor and premotor cortices. Thus, there are major species differences in cortical dopamine projections.
The posterior hypothalamus. These dopaminergic cells (group A11) project to the spinal cord, and their function is not well established. There is some evidence that pathology in this area plays a role in restless legs syndrome, a condition in which people have difficulty sleeping due to an overwhelming compulsion to constantly move parts of the body, especially the legs.
The arcuate nucleus (cell group A12) and periventricular nucleus (cell group A14) of the hypothalamus. An important projection from these dopaminergic neurons goes to thepituitary gland, where it influences the secretion of the hormone prolactin. Dopamine is the primary neuroendocrine inhibitor of the secretion of prolactin from the anterior pituitary gland. Dopamine produced by neurons in the arcuate nucleus is secreted into the hypothalamo-hypophysial blood vessels of the median eminence, which supply thepituitary gland. The lactotrope cells that produce prolactin, in the absence of dopamine, secrete prolactin continuously; dopamine inhibits this secretion. Thus, in the context of regulating prolactin secretion, dopamine is occasionally called prolactin-inhibiting factor (PIF), prolactin-inhibiting hormone (PIH), or prolactostatin.
The zona incerta. These cells (group A13) project to several areas of the hypothalamus, and participate in the control of gonadotropin-releasing hormone, which is necessary to activate the development of reproductive systems that occurs following puberty, both in males and females.
An additional group of dopamine-secreting neurons are located in the retina of the eye. These neurons are amacrine cells, meaning that they have no axons. They release dopamine into the extracellular medium, and are specifically active during daylight hours, becoming silent at night. This retinal dopamine acts to enhance the activity of cone cellsin the retina while suppressing rod cells — the result is to increase sensitivity to color and contrast during bright light conditions, at the cost of reduced sensitivity when the light is dim.
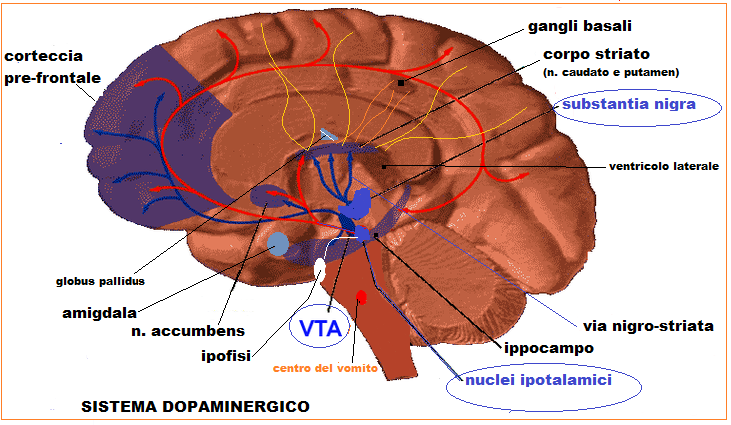
_Figura 1 -centri di produzione dopamina
1.Schultz W (2007). Multiple dopamine functions at different time courses
2.A. Dahlström and K. Fuxe (1964). Evidence for the existence of monoamine-containing neurons in the central nervous system. I. Demonstration of monoamines in the cell bodies of brain stem neurons
3.Björklund A, Dunnett SB (May 2007). Dopamine neuron systems in the brain: an update(07)00067-7?cc=y
4.Christine CW, Aminoff MJ (September 2004. Clinical differentiation of parkinsonian syndromes: prognostic and therapeutic relevance(04)00362-6/fulltext
5.DeLong M, Wichmann T (April 2010). Changing views of basal ganglia circuits and circuit disorders
6.Robbins TW, Arnsten AF. (2009). The neuropsychopharmacology of fronto-executive function: monoaminergic modulation
7.Paulus W, Schomburg ED (June 2006). Dopamine and the spinal cord in restless legs syndrome: does spinal cord physiology reveal a basis for augmentation?
8.Ben-Jonathan N, Hnasko R (2001)."Dopamine as a Prolactin (PRL) Inhibitor":http://press.endocrine.org/doi/abs/10.1210/edrv.22.6.0451
9.Witkovsky P (January 2004). Dopamine and retinal function
PHATOPHYSIOLOGY
The primary symptoms of Parkinson's disease result from greatly reduced activity of dopamine-secreting cells caused by cell death in the pars compacta region of the substantia nigra. There are five major pathways in the brain connecting other brain areas with the basal ganglia. These are known as the motor, oculo-motor, associative, limbic and orbitofrontal circuits, with names indicating the main projection area of each circuit. All of them are affected in PD, and their disruption explains many of the symptoms of the disease since these circuits are involved in a wide variety of functions including movement, attention and learning. Scientifically, the motor circuit has been examined the most intensively.
Dopamine deficiency leads not only to parkinsonian motor signs but also to anxiety, impaired cognition, or depressed mood.
The discovery of dopamine D3 receptors (D3Rs) and their anatomic localization to brain regions associated with limbic and prefrontal cortex stimulated great scientific interest. These regions include ventral pallidum, amygdala, ventral tegmental area, the islands of Calleja, and striatal striosomes. This anatomy makes plausible the hypothesis that activating D3Rs may preferentially affect the limbic or prefrontal cortical regions previously implicated in dopamine-influenced complex behavior. However, an agonist applied to a receptor in one brain region (e.g., putamen) can affect neuronal function in remote brain regions (e.g., motor cortex). Behavioral disinhibition observed with dopaminomimetic agents (in PD therapy) may be mediated in part by an inhibitory influence of D3R activation on orbitofrontal cortex.
Obeso JA, Rodríguez-Oroz MC, Benitez-Temino B, et al. (2008). Functional organization of the basal ganglia: therapeutic implications for Parkinson's disease
Kevin J. Black,*†‡§ Tamara Hershey,*Jonathan M. Koller,* Tom O. Videen,†‡ Mark A. Mintun,*‡ Joseph L. Price,∥ andJoel S. Perlmutter†‡∥ (2002) “A possible substrate for dopamine-related changes in mood and behavior, Prefrontal and limbic effects of a D3-preferring dopamine agonist”:http://www.ncbi.nlm.nih.gov/pmc/articles/PMC139278/

_Figura 2 - trasduzione del segnale mediante recettori D1, D2, D3
The D1 receptors facilitate adenyl cyclase, whereas the D2 receptors (like D3) inhibit this second messenger. These processes change the protein-synthesizing capabilities of the cell. D3 is a G-protein coupled receptor (GPCR). In the simplest conceptualization, GPCRs work as a switch. When a G-protein is attached to the cellular side of a GPCR, the GPCR exists in high-affinity for its ligand, but when the G-protein detaches from the GPCR, the GPCR has very low affinity for DA. D3receptors are unique among the D2-like receptors, exhibiting sustained high affinity for DA (>20-fold higher than D2 receptors), suggesting that D3 receptors in vivo may be occupied by endogenous DA for extended periods of time, leading to high spontaneous activation of D3 receptors.
Vanhauwe JF, Josson K, Luyten WH, Driessen AJ, Leysen JE (2000) G-protein sensitivity of ligand binding to human dopamine D (2) and D(3) receptors expressed in Escherichia coli: clues for a constrained D(3) receptor structure
Richtand NM, Woods SC, Berger SP, Strakowski SM (2001) D3 dopamine receptor, behavioral sensitization, and psychosis.Neurosci Biobehav Rev

_Figura - recettore D3
PARKINSON'S DISEASE AND CREATIVITY
A creative work is not the result of a single cognitive process, of neuronal activity in one isolated cerebral area, or of a unique mental faculty, but of the interaction of multiple cognitive processes supported by a large network of multiple cerebral areas. The neurotransmitter dopamine plays a crucial role in this complex interaction. Drugs that can lead to addiction induce a large release of dopamine in the mesolimbic pathway, directly for amphetamine and cocaine by blocking dopamine reuptake, and indirectly for other psychoactive drugs such as alcohol. The dopaminergic mesolimbic pathway is involved in the control of motivational, reward, and reinforcement processes and contributes to dependence and addiction. In Parkinson’s disease (PD), depletion of the substantia nigra and the ventral tegmental area causes low dopamine concentration in the brain and cognitive impairment diminishes flexibility processes essential to creativity. To Lakke’s surprise, artistic activity persisted in PD, and in the case of several patients was awakened, from the time of disease onset. Many case reports confirm exacerbation or revelation of creative art work in PD patients treated by dopamine replacement therapy (DRT) and especially dopamine agonists. Creative patients have higher doses of dopamine agonists than controls presenting the same motor severity. Creativity appears with, or is exacerbated by, dopaminergic treatment, and decreases when dopaminergic treatment is reduced, in the context of STN stimulation. Dopamine agonists have a more selective affinity with D3 dopaminergic receptors, which are more highly represented in the mesolimbic system, than l-DOPA which has a more diffuse and balanced action on all dopaminergic receptors. Overstimulation of mesolimbic dopaminergic systems seems to facilitate the drive to create in PD patients who possess a certain “creative intelligence,” in a non-specific way, via the enhancement of all directed behaviors with a reward component. Dopaminergic treatment in PD induces ultra-rapid mood cycles, with euphoric/dysphoric oscillations, constituting a favorable ground for creativity, as it is the case in bipolar disorder. While behavioral addictions and impulse control disorders are well-known potentially devastating side effects, creativity can generally be considered as a highly beneficial side effect of dopamine agonists.
Eugénie Lhommée, Alina Batir, [...], and Paul Krack (2014). Dopamine and the Biology of Creativity: Lessons from Parkinson’s Disease

_Figura 4 - opera di una paziente affetta da Parkinson

_Figura 5 - opera di una paziente affetta da Parkinson