Synthesis from GTP
Every cell is able to synthesize BH4 (tetrahydrobiopterin) from GTP.

Biosynthesis of tetrahydrobiopterin starts from guanosine triphosphate by the action of guanosine triphosphate cyclohydrolase I, which yields the first intermediate, 7,8-dihydroneopterin triphosphate. This compound is then converted by subsequent enzymes, 6-pyruvoyl tetrahydropterin synthase and sepiapterin reductase, to tetrahydrobiopterin, the biologically active metabolite. Cytokines such as gamma-interferon or tumor necrosis factor-alpha strongly stimulate the activity of guanosine triphosphate cyclohydrolase I in murine and human cells, yielding a potentiation of intracellular tetrahydrobiopterin concentrations. In human cells, particularly in human monocytes and macrophages, the low activity of 6-pyruvoyl tetrahydropterin synthase leads to the additional accumulation of neopterin derivatives, which leak from the cells after dephosphorylation and are found increased in body fluids of humans with diseases challenging cell-mediated immunity. A functional role for the stimulation of tetrahydrobiopterin biosynthesis by cytokines is the formation of a limiting cofactor required for the enzymatic conversion of L-arginine to citrulline and nitric oxide. (Tetrahydrobiopterin and cytokines. 1993)
A double edge sword:6-pyruvoyl tetrahydropterin reductase
BH4 synthesis is stimulated by Ascorbic Acid (AA).
Effect of vitamin C on the availability of tetrahydrobiopterin in human endothelial cells. 2001
BH4 is an essential cofactor for the synthesis of:
- dopamine
- noradrenaline and adrenaline
- serotonine
- NO (Nitric Oxide)
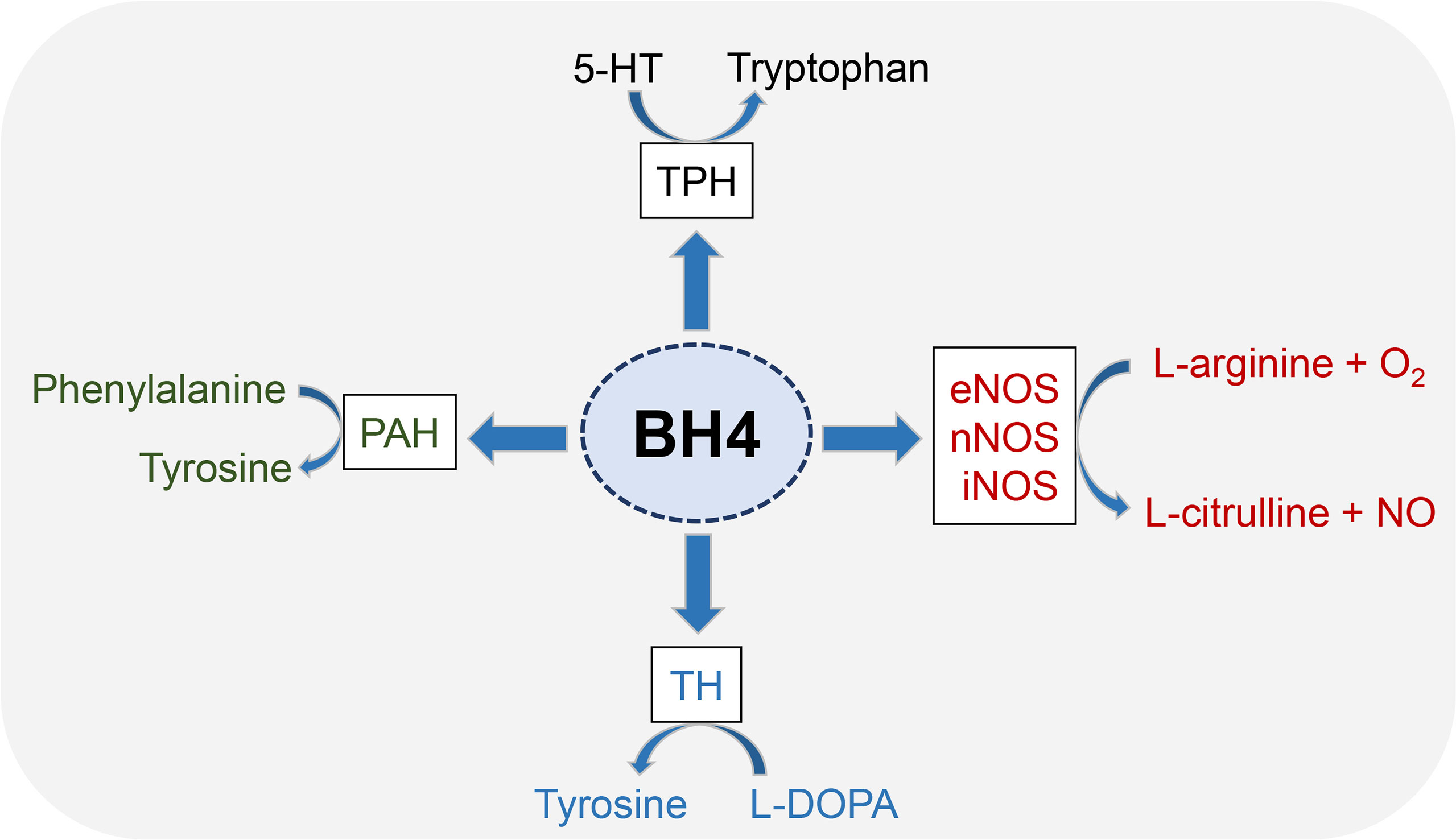

BH4 acts as a redox agent shuttling reducing equivalents from NADPH to the specific substrate (eg in hydroxylase reactions).

Working hypothesis: BH4 main role is to be the pace-maker of NO and neurotransmitters synthesis
Role of BH4 in NOS activity
BH4 is required for the expression of NOS activity, but it has been a source of considerable controversy as to what roles it might be playing, and in particular, whether it plays a redox role in the reaction mechanism.
The table below shows the diverse range of functions proposed for BH 4 in NOS. Intriguingly none of these involve the two-electron BH2/BH4 cycle that is the 'classical' role for BH 4 in aromatic amino acid hydroxylases.
- Promotes coupling of NADPH oxidation to NO synthesis and inhibits superoxide and hydrogen peroxide formation;
- Promotes dimer formation and increases tightness/stability of dimer (e.g. to SDS);
- Allosteric binding effects: on subsequent arginine binding and on the binding of a second molecule of BH4;
- Modifies haem environment (low spin to high spin);
- Protects against inactivation, autoinactivation;
- Redox active in catalysis.
BH4 binds to the haem-containing domain of NOS, interacting with the haem propionate and thus binding immediately adjacent to the active site where L-arginine is bound, and also forming part of the NOS dimer interface.
Regulation of pteridine-requiring enzymes by the cofactor tetrahydrobiopterin.
Role of BH4 in neurotransmitters synthesis
BH4 synthesis Regulation
 Glucocorticoids BH4
Glucocorticoids BH4
BH2 reduction to BH4
BH4/BH2 recycling
Kegg Pathways
Critical Role for Tetrahydrobiopterin Recycling by Dihydrofolate Reductase in Regulation of Endothelial Nitric-oxide Synthase Coupling 2009
The availability of BH4 versus the abundance of 7,8-dihydrobiopterin (BH2) drive the activity of BH4-dependent enzymes.


BH2 reduction rate depends on activity of
- DHFR (Dihydrofolate Reductase)
- angiotensin II down-regulated DHFR expression
- DHPR (Dihydropteridine Reductase)
- MTHFR ?? . Due to the high prevalence of MTHFR polymorphisms with reduced activity a large variability of BH2 reduction to BH4 can be expected.
- Characterization of the dihydropterin reductase activity of pig liver methylenetetrahydrofolate reductase. J Biol Chem. 1980 Jul 10;255(13):6014-7. Matthews RG, Kaufman S.
- Pig liver methylenetetrahydrofolate reductase catalyzes the reduction of quinonoid dihydropterins in vitro. Either NADPH or methyltetrahydrofolate can serve as the electron donor. Methylenetetrahydrofolate reductase can also suppor phenylalanine hydroxylation in vitro by regeneration of the tetrahydropterin cofactor. These results lend support to the proposal that reduction of methylenetetrahydrofolate proceeds by tautomerization of the 5-iminium cation to form quinonoid 5-methyldihydrofolate, which is then reduced to methyltetrahydrofolate (Matthews, R. G., and Haywood, B. J. (1979) Biochemistry 18, 4845-4851). Under Vmax conditions, the turnover numbers for the NADPH-linked reductions of the quinonoid forms of 6,7-dimethyldihydropterin, dihydrobiopterin, and dihydrofolate are all about the same as that for the reduction of methylenetetrahydrofolate. The Km values for racemic mixtures of the same quinonoid acceptors are 40, 30, and 20 microM, respectively, while the Km for (6R,S)methylenetetrahydrofolate is 20 microM at pH 7.2 in phosphate buffer. The reduction of quinonoid dihydropterins is inhibited by adenosylmethionine and dihydropteroylhexaglutamate, which are known to modulate methylenetretrahydrofolate reductase activity.
- The effect of tetrahydrofolate on tetrahydrobiopterin metabolism. J Ment Defic Res. 1986 Jun;30 ( Pt 2):17.9-83 Hamon CG, Blair JA, Barford PA.

Open Question: BH2 isoforms are in equilibium or derive from different chemical reaction?
5-Methyltetrahydrofolate and vitamin B12 appear to be required for the biosynthesis of tetrahydrobiopterin. A deficiency of either could be sufficient to bring about neurological change which can be corrected by reversing the deficiency. Patients with senile dementia could possibly be benefited by the administration of 5-methyltetrahydrofolate.
H2O2 and BH 4
Hydrogen peroxide stimulates tetrahydrobiopterin synthesis through the induction of GTP-cyclohydrolase I and increases nitric oxide synthase activity in vascular endothelial cells 2003
Lack of BH 4 leads to increased production of H2O2 by NOS; a cyclic regulatory pathway
The piruvoyl-reductase step is catalized by the enzyme aldose reductase (W) belonging to the polyol pathway
Human carbonyl and aldose reductases: new catalytic functions in tetrahydrobiopterin biosynthesis. 1991
H2O2 and BH 4
BH4 and cell proliferation
Tetrahydrobiopterin levels regulate endothelial cell proliferation. 2001
Neopterin and osteoporosis (and Rheumatoid Arthritis)
Kynurenine and neopterin levels in patients with rheumatoid arthritis and osteoporosis during drug treatment. 2003
Kynurenine pathway metabolism in patients with osteoporosis after 2 years of drug treatment. 2006

Genetic Diseases linked to defects of BH 4 synthesis
Analysis of Neurotransmitters”, Simon Heales
Coronary endothelial dysfunction in the insulin-resistant state is linked to abnormal pteridine metabolism and vascular oxidative stress 2001
Furthermore, not only decrements of the plasma BH4/7,8-BH2 ratio and DHPR but also enhancement of oxidative stress and defects of antioxidant defenses were observed in these subjects. Conversely, ACh-induced vasodilation was positively correlated with insulin sensitivity, BH4/7,8-BH2 ratio and DHPR activity. Thus, in addition to overproduction of LPO in the coronary artery, decreased BH4/7,8-BH2 ratio also appeared to be an independent predictor of impaired epicardial vasodilator responses to ACh, suggesting that these factors exerted additional adverse effects on the endothelial function of the coronary artery.
Glucocorticoid-Induced Hypertension and Tetrahydrobiopterin (BH4), a
Common Cofactor for the Production of Vasoactive Molecules 2005
Recent research has focused on the effects of excess glucocorticoids on BH4 biosynthesis because reduced BH4 cofactor levels can alter the production of NO, serotonin, and catecholamines by NO synthase and the aromatic amino acid hydroxylases. This review will focus on the mechanisms and consequences of excess glucocorticoids on the BH4 biosynthesis pathway and the enzymes that utilize BH4 as a cofactor.
Mechanisms underlying recoupling of eNOS by HMG-CoA reductase inhibition in a rat model of streptozotocin-induced diabetes mellitus 2009
HMG-CoA reductase inhibitors have been shown to upregulate GTP cyclohydrolase I (GTPCH-I), the key enzyme for tetrahydrobiopterin de novo synthesis and to normalize tetrahydrobiopterin levels in hyperglycemic endothelial cells.
BH4 replacement therapy
Sapropterina
Scheda Tecnica del Seguente Nuovo Medicinale Orfano Autorizzato in Data
02 Dicembre 2008 dalla Commissione Europea all'Autorizzazione all'Immissione
in Commercio :
- Farmaco : Kuvan (Compresse Solubili)
- Principio Attivo : Sapropterina Dicloridrato
La Sapropterina e' una Versione Sintetica della Molecola 6R-BH4 Esistente in
Natura, la Quale e' un Cofattore dell'Idrossilasi per Fenilalanina e Triptofano
- ATC : A16AX07
- Gruppo Farmacoterapeutico : Prodotti Vari dell' Apparato Gastrointestinale e
Metabolismo
- Indicazione Terapeutica : Trattamento dell'Iperfenilalaninemia (HPA) in Pazienti
Adulti e Pediatrici al di Sopra dei Quattro Anni di Eta' Affetti da Fenilchetonuria
(PKU) che hanno Mostrato una Risposta a tale Trattamento.
E' Anche Indicato per il Trattamento dell'Iperfenilalalinemia (HPA) in Pazienti
Adulti e Pediatrici con Carenza di Tetraidrobiopterina (BH4) che hanno Mostrato
una Risposta al Trattamento
Folate nutritional genetics and risk for hypertension in an elderly population sample. 2009
J Nutrigenet Nutrigenomics. 2009;2(1):1-8. Epub 2008 Oct 8.
Ng X, Boyd L, Dufficy L, Naumovski N, Blades B, Travers C, Lewis P, Sturm J, Yates Z, Townley-Jones M, Roach P, Veysey M, Lucock M.
School of Environmental and Life Sciences, University of Newcastle, Ourimbah, NSW, Australia.
Abstract
BACKGROUND/AIMS: 118 elderly participants (65-90 years) were assessed for any relationship between folate, related genes and hypertension. METHODS: Six B-vitamin-related SNPs were genotyped in 80 normotensive and 38 hypertensive subjects. RESULTS: Of six polymorphisms (677C>T-MTHFR, 1298A>C-MTHFR, 80G>A-RFC, 2756A>G-MS, 66A>G- MSR, 19bpDHFR and 1561C>T-GCPII), only 677C>T-MTHFR was a significant risk for hypertension: OR 1.89; 95% CI 1.07-3.32 (chi2 p = 0.038). Additionally, hypertensive subjects had a significantly lower intake of dietary folate than normotensive individuals (p = 0.0221), although this did not markedly alter blood metabolite levels. Several significant linear associations between dietary folate and related blood metabolites were found in normotensive subjects (p < 0.001 for Hcy, red cell and serum folate) and were as predicted on an a priori basis -- generally weaker associations existed in hypertensive subjects (p < 0.05 for serum folate). This was true for data examined collectively or by genotype. Multiple-regression analysis for diastolic or systolic blood pressure showed significant interaction for gender and folate intake (p = 0.014 and 0.019, respectively). In both cases this interaction occurred only in females, with higher folate intake associated with decreased blood pressure. Regressing diastolic blood pressure and 677C>T-MTHFR genotype showed significance (males; p = 0.032) and borderline significance (all subjects). CONCLUSION: Dietary folate and 677C>T-MTHFR genotype may modify blood pressure. Copyright 2008 S. Karger AG, Basel.
Aldose reductase deficiency protects from autoimmune- and endotoxin-induced uveitis in mice. 2012
- Further, expression of inflammatory marker proteins such as inducible nitric oxide synthase (iNOS), cyclooxygenase (COX)-2, tumor necrosis factor (TNF)-α, and vascular cell adhesion molecule (VCAM)-1 was increased in the WT-EIU mouse eyes but not in the ARKO-EIU eyes.
Schirks Laboratories
Pteridine Price List Schirks
Neopterin and inflammation
Serum Levels of Neopterin, Galectin-3, Tissue Necrosis Factor Alpha, and Interleukin-10 During the Interictal and Attack Periods of Patients with Migraine, 2023
Abstract
Objective: This study aimed to determine the serum levels of neopterin, galectin-3, tissue necrosis factor alpha, and interleukin-10 (IL-10) during the interictal and attack periods of patients with migraine. It is hoped that the findings will contribute to the literature on the theory of neurogenic inflammation.
Materials and Methods: Eighty patients with migraine (40 attacks and 40 interictal periods) and 30 healthy volunteers were included in the study. Blood samples were taken from the people participating in the study, and serum was obtained under the appropriate conditions. The serum levels of all the parameters were determined using a ELISA kit (Coon Koon, Shanghai, China). The difference between the groups was evaluated using appropriate statistical analysis methods.
Results: Galectin-3 and neopterin levels were higher during the attack period compared with the interictal period, and IL-10 and galectin-3 levels were higher during the attack period than in the control group. There was no significant difference in any parameter between the control and interictal period groups. There was a correlation between galectin-3 and neopterin levels in all groups (r = 0.598, P < 0.001).
Conclusion: The levels of pro-inflammatory molecules, such as galectin-3 and neopterin, were found to be higher in patients with migraine during attacks, which is consistent with the theory of neurogenic inflammation. It is thought that the increase in IL-10 levels during the attack occurs to limit inflammation. Since the tetrahydrobiopterin pathway plays a role in inflammatory and neuropathic pain, it is thought that high neopterin levels during an attack may be associated with migraine headaches.