endogenous+digoxin+synthesis
HMG+CoA+reductase+Digoxin
HMG+CoA+reductase+endogenous+Digoxin
HMG+CoA+reductase+endogenous+ouabain
HMG+CoA+reductase+Endogenous+digitalis-like+factors
down+syndrome+and++Endogenous+digitalis-like+factors

Cardiac glycosides with target at direct and indirect interactions with nuclear receptors, 2020

Endogenous Ouabain: Upregulation of Steroidogenic Genes in Hypertensive Hypothalamus but Not Adrenal, 2005


Emergence of Cardiac Glycosides as Potential Drugs: Current and Future Scope for Cancer Therapeutics, 2021
Disambiguation: CTS cardiotonic steroids or EDLS Endogenous digitalis-like Substance or HIF Hypothalamic Inhibitory Factor
Endogenous digitalis: pathophysiologic roles and therapeutic applications 2008
Nature Reviews Nephrology 4, 378-392 (July 2008) | doi:10.1038/ncpneph0848
Alexei Y Bagrov & Joseph I Shapiro
Abstract

Neurosteroids and 5-alpha-reductase
Endogenous digitalis-like factors, also called cardiotonic steroids, have been thought for nearly half a century to have important roles in health and disease. The endogenous cardiotonic steroids ouabain and marinobufagenin have been identified in humans, and an effector mechanism has been delineated by which these hormones signal through the sodium/potassium-transporting ATPase. These findings have increased interest in this field substantially. Although cardiotonic steroids were first considered important in the regulation of renal sodium transport and arterial pressure, subsequent work has implicated these hormones in the control of cell growth, apoptosis and fibrosis, among other processes. This Review focuses on the role of endogenous cardiotonic steroids in the pathophysiology of essential hypertension, congestive heart failure, end-stage renal disease and pre-eclampsia. We also discuss potential therapeutic strategies that have emerged as a result of the increased understanding of the regulation and actions of cardiotonic steroids.
- Adrenocorticotropic hormone
- angiotensin II
- vasopressin
- phenylephrine
stimulate the release of ouabain from the adrenal cortex in vitro
the sites of ouabain production in mammals.
- The adrenal cortex
- hypothalamus
Endogenous cardiac glycosides, a new class of steroid hormones. 2002


Fig. 2. Hormonal control of the release of endogenous ouabain from zona fasciculata cells of the adrenal cortex by hypernatriaemia, hypoxia, and physical exercise. The scheme represents a compilation of the findings that ouabain levels are increased in patients with low-renin hypertension [50], in hypoxemia [39,40], and during physical exercise [42]. In adrenocortical cells, ouabain is released by phenylephrine and angiotensin II, and its action is blocked by the AT2 inhibitor PD123319 [36–38]. The ACE inhibitor prazolol and the β-blocker atenolol abolish the exercise-dependent rise in ouabain levels in dogs [42]. Digoxin counteracts the ouabain-dependent rise in blood pressure in rats [48,57,59] and lowers renin levels in humans [86]. It has been shown that ouabain and angiotensin II stimulate the release and synthesis of catecholamines and that catecholamines stimulate the release of renin. Increased concentrations of Na+ increase the synthesis of angiotensin converting enzyme (ACE) [107]. Stimulation of ouabain release by adrenocorticotropin has been observed in tissue culture [36] and in vivo in the rat [44], but not in dog and man [42,43].
Much information has now accumulated indicating that the plant toxin ouabain is a mammalian steroid hormone [18,20–22] that is under the rapid control of catecholamines and/or angiotensin [28,34,36–38,42]. The picture is emerging that increased plasma Na+ concentrations [47], hypoxemia [39,40], and physical exercise [42] stimulate the release of endogenous ouabain and possibly other cardiotonic steroids from hypothalamic and adrenocortical cells. The increase in plasma ouabain leads in turn to an increase in blood pressure (Fig. 2). This rise is not associated with plasma renin activity [106]. The release of ouabain from zona fasciculata cells of the adrenal gland is stimulated via the angiotensin and adrenergic systems, which are intimately interconnected [107]. Increased endogenous ouabain concentrations are associated with about 50% of hypertension cases diagnosed in Caucasians with low renin hypertension [50]. According to Hamlyn et al., elevated levels of ouabain primarily affect the central nervous system and activate angiotensin II-dependent pathways in the brain that mediate sympathetic nerve activity [16]. Circulating ouabain and its interplay with the tissue renin-angiotensin and sympathetic nervous systems might also induce functional and structural cardiovascular changes by peripheral mechanisms [16].

Indian J Exp Biol. 2000 May;38(5):438-46.Links
Isoprenoid pathway and free radical generation and damage in neuropsychiatric disorders
Ravikumar A, Arun P, Devi KV, Augustine J, Kurup PA.
Department of Neurology, Medical College, Thiruvananthapuram 695 011, India.
Two substances which are products of the isoprenoid pathway, can participate in lipid peroxidation. One is digoxin, which by inhibiting membrane Na(+)-K+ ATPase, causes increase in intracellular Ca2+ and depletion of intracellular Mg2+, both effects contributing to increase in lipid peroxidation. Ubiquinone, another products of the pathway is a powerful membrane antioxidant and its deficiency can also result in defective electron transport and generation of reactive oxygen species. In view of this and also in the light of some preliminary reports on alteration in lipid peroxidation in neuropsychiatric disorders, a study was undertaken on the following aspects in some of these disorders (primary generalised epilepsy, schizophrenia, multiple sclerosis, Parkinson's disease and CNS glioma)--1) concentration of digoxin, ubiquinone, activity of HMG CoA reductase and RBC membrane Na(+)-K+ ATPase 2) activity of enzymes involved in free radical scavenging 3) parameters of lipid peroxidation and 4) antioxidant status. The result obtained indicates an increase in the concentration of digoxin and activity of HMG CoA reductase, decrease in ubiquinone levels and in the activity of membrane Na(+)-K+ ATPase. There is increased lipid peroxidation as evidenced from the increase in the concentration of MDA, conjugated dienes, hydroperoxides and NO with decreased antioxidant protection as indicated by decrease in ubiquinone, vit E and reduced glutathione in schizophrenia, Parkinson's disease and CNS glioma. The activity of enzymes involved in free radical scavenging like SOD, catalase, glutathione peroxidase and glutathione reductase is decreased in the above diseases. However, there is no evidence of any increase in lipid peroxidation in epilepsy or MS. The role of increased operation of the isoprenoid pathway as evidenced by alteration in the concentration of digoxin and ubiquinone in the generation of free radicals and protection against them in these disorders is discussed.
Isoprenoid pathway related cascade in multiple myeloma., 2003
HYPOTHALAMIC DIGOXIN, HEMISPHERIC CHEMICAL DOMINANCE, AND ONCOGENESIS: EVIDENCE FROM MULTIPLE MYELOMA 2003

Clinical and therapeutic significance of the Na+,K+ pump*
angiotensin II stimulated release of aldosterone, cortisol, and a CS that had similar polarity to ouabain and was recognized by a ouabain antibody (12, 30). Stimulation of steroid secretion by angiotensin II was dose dependent for all three steroids: half-maximally effective concentrations of angiotensin II were 0.31–0.38 nM/l for secretions of aldosterone and cortisol and 2.3 nM/l for ouabain-like CS.
ref:
[22] J.M. Hamlyn, Z. Lu, P. Manunta, J.H. Ludens, K. Kimura and J.R. Shah et al., Observations on the nature, biosynthesis, secretion and significance of endogenous ouabain, Clin Expl Hypertens 20 (1998), pp. 523–533. View Record in Scopus | Cited By in Scopus (80)
[23] Y. Komiyama, N. Nishimura, M. Munakata, T. Mori, K. Okuda and N. Nishino et al., Identification of endogenous ouabain in culture supernatant of PC12 cells, J Hypertens 19 (2001), pp. 229–236. Full Text via CrossRef | View Record in Scopus | Cited By in Scopus (17)
[24] A. Perrin, B. Brasmes, E.M. Chambaz and G. Defaye, Bovine adrenocortical cells in culture synthesize an ouabain-like compound, Mol Cell Endocrinol 26 (1997), pp. 7–15. Abstract | PDF (687 K) | View Record in Scopus | Cited By in Scopus (37)
..elicited by the interaction of ouabain with Na(+)/K(+)-ATPase, and are...genes, and stimulation of protein synthesis and myocyte hypertrophy. For...function of Na+/K+-ATPase are the endogenous ouabain-like hormones, and changes in...
 Papers HMG CoA reductase Digoxin
Papers HMG CoA reductase Digoxin

Fig. 3: Signaling pathways of cardiotonic steroids a Activation of NFκB via slow Ca++ oscillations following modification of Ca++ release from endoplasmic reticulum. b Activation of mitochondrial ROS via src and EGF-R. c Activation of nuclear gene programs via scr and EGF-R. PKC protein kinase C
Image Text (High Precision): antiproliferation apoptosi apoptosis cardiotonic cell endoplasmic ouabain oxygen proliferation reactive reticulum species steroids
fulltext
Salt restriction in kidney disease—a missed therapeutic opportunity?
Synthesis

Hypothalamic digoxin and isoprenoid pathway dysfunction relation to alcoholic addiction, alcoholic cirrhosis, and acquired hepatocerebral degeneration--relation to hemispheric chemical dominance, 2003, FULLTEXT
Hypothalamic digoxin and isoprenoid pathway dysfunction relation to alcoholic addiction, alcoholic cirrhosis, and acquired hepatocerebral degeneration--relation to hemispheric chemical dominance, 2003
The biosynthesis of EO involves, cholesterol side-chain cleavage (CYP11A1), 3-beta-hydroxysteroid dehydrogenase (HSD3B) with sequential metabolism of pregnenolone and progesterone.
Hypothetical biosynthetic pathway for conversion of cholesterol to endogenous cardiac glycosides. Relevant human genes coding for pathway enzymes are identified by Locus Link numbers and gene symbol. Rat orthologue genes in bold were found upregulated by rat microarray analysis. Rat orthologues italicized and underlined were not available on the CodeLink microarrays. Structures show plant ouabain and candidate precursor molecule.


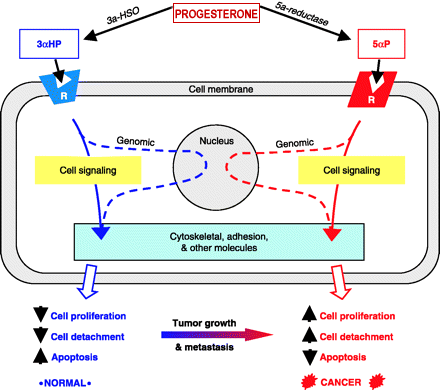
14 beta-Hydroxyprogesterone binds to the digitalis receptor, inhibits the sodium pump and enhances cardiac contractility. 1998
 Papers endogenous ouabain synthesis progesterone
Papers endogenous ouabain synthesis progesterone
 Papers digoxin ouabain antagonist
Papers digoxin ouabain antagonist
Contacts and cooperation between cells depend on the hormone ouabain 2006

Role of Na+,K+-ATPase and its modulation by ouabain. (A) The enzyme consists of a catalytic α-subunit and a β-subunit that has a long extracellular domain with adhesive properties (23). (B) Because of β/β adhesiveness the enzyme stabilizes at the intercellular space, which is sealed at the outermost end by the TJ and opens toward the interstitial side. Because of the firmness of the α/β union, the whole enzyme resides at the borders of the intercellular space. © Monolayers of MDCK in transverse optical sections show the Na+,K+-ATPase (green) at the cell–cell borders but not at the apical or basal domains. Nuclei were stained with propidium iodine (red). (D) Pumps located in the lateral cell border facing the intercellular space lower the Na+ concentration in the cytoplasm, and the gradient of this ion across the apical border acts as a driving force for cotransporters and countertransporters (brown and gray circles) translocating H+, K+, Ca2+, sugars, amino acids, etc. (E) Ouabain (pink circles) causes endocytosis of pumps as in Fig. 4 B, thereby diminishing the driving force for translocating mechanisms and decreasing the flux of nutrients. (F) When one of the cells is a ouabain-resistant one ®, cell–cell attachment molecules are not retrieved from its membrane or from the membrane of its neighbor W, ouabain-binding pumps of the W cell are not endocytosed (Fig. 4 C), and cotransporters remain in operation because the Na+ gradient across the apical membrane does not dissipate. Ionic balance of the W cells is maintained by the ouabain-resistant pumps of R cells (14) as well as the presence of gap junctions, which, because of the enhancement of connexin 32 (Fig. 3), increases cell–cell communication (Fig. 2).
Calcium, ATP, and ROS: a mitochondrial love-hate triangle 2004
Endogenous ouabain-like factor (OLF) secretion is modulated by nicotinic mechanisms in rat adrenocortical cells. 2007
Life Sci. 2004 Mar 12;74(17):2111-28.
Göõz M, Tóth M, Vakkuri O, Göõz P, Smolka AJ, de Châtel R, Szalay KS.
This study tested the hypothesis that rat adrenocortical secretion of endogenous ouabain-like factor (OLF) is regulated by nicotinic mechanisms. OLF secreted by dispersed cell suspensions of zona glomerulosa (ZG) and fasciculata/reticularis (ZFR) cells was found to co-elute with authentic ouabain by reverse phase HPLC; OLF concentrations in cell supernatants were measured by radioimmunoassay. Nicotine (10(-6) - 10(-3) M) stimulated significant OLF secretion in rat adrenocortical cells. Acetylcholine (10(-7) - 10(-4) M) and eserine (10(-7) - 10(-3) M) stimulated OLF secretion in ZG cells at lower concentrations and stimulated at higher concentrations. Acetylcholine had no effect on ZFR secretion of OLF, but eserine stimulated OLF secretion. ACTH (10(-8) M) strongly potentiated the OLF stimulatory effect of nicotine in ZG cells; however significant interactions between nicotine and ACTH or angiotensin II on OLF secretion in ZFR cells were not apparent. The ganglionic blockers hexamethonium and mecamylamine further potentiated the effect of nicotine, implicating nicotinic acetylcholine receptors (nAChRs) in regulation of OLF secretion. The alpha7-receptor antagonist methyllycaconitine (MLA) dose-dependently inhibited the effect of nicotine in the ZG cells, and in ZFR cells MLA potentiated nicotine-induced OLF secretion. These data suggest that nicotinic regulation may underlie OLF secretion by rat adrenocortical cells, and strongly suggest presence of functional nicotinic acetylcholine receptors on these cells.
Antagonistic effect of ouabain and digoxin
Endogenous and exogenous cardiac glycosides and their mechanisms of action. 2007
Am J Cardiovasc Drugs. 2007;7(3):173-89.
Schoner W, Scheiner-Bobis G.
Cardiac glycosides have been used for decades to treat congestive heart failure. The recent identification of cardiotonic steroids such as ouabain, digoxin, marinobufagenin, and telocinobufagin in blood plasma, adrenal glands, and hypothalamus of mammals led to exciting new perspectives in the pathology of heart failure and arterial hypertension. Biosynthesis of ouabain and digoxin occurs in adrenal glands and is under the control of angiotensin II, endothelin, and epinephrine released from cells of the midbrain upon stimulation of brain areas sensing cerebrospinal Na(+) concentration and, apparently, the body's K(+) content. Rapid changes of endogenous ouabain upon physical exercise may favor the economy of the heart by a rise of intracellular Ca(2)(+) levels in cardiac and atrial muscle cells. According to the sodium pump lag hypothesis, this may be accomplished by partial inhibition of the sodium pump and Ca(2+) influx via the Na(+)/Ca(2+) exchanger working in reverse mode or via activation of the Na(+)/K(+)-ATPase signalosome complex, generating intracellular calcium oscillations, reactive oxygen species, and gene activation via nuclear factor-kappaB or extracellular signal-regulated kinases 1 and 2. Elevated concentrations of endogenous ouabain and marinobufagenin in the subnanomolar concentration range were found to stimulate proliferation and differentiation of cardiac and smooth muscle cells. They may have a primary role in the development of cardiac dysfunction and failure because (i) offspring of hypertensive patients evidently inherit elevated plasma concentrations of endogenous ouabain; (ii) such elevated concentrations correlate positively with cardiac dysfunction, hypertrophy, and arterial hypertension; (iii) about 40% of Europeans with uncomplicated essential hypertension show increased concentrations of endogenous ouabain associated with reduced heart rate and cardiac hypertrophy; (iv) in patients with advanced arterial hypertension, circulating levels of endogenous ouabain correlate with BP and total peripheral resistance; (v) among patients with idiopathic dilated cardiomyopathy, high circulating levels of endogenous ouabain and marinobufagenin identify those individuals who are predisposed to progressing more rapidly to heart failure, suggesting that endogenous ouabain (and marinobufagenin) may contribute to toxicity upon digoxin therapy.
In contrast to endogenous ouabain, endogenous marinobufagenin may act as a natriuretic substance as well.
- It shows a higher affinity for the ouabain-insensitive alpha(1) isoform of Na(+)/K(+)-ATPase of rat kidney tubular cells and its levels are increased in volume expansion and pre-eclampsia.
- Digoxin, which is synthesized in adrenal glands, seems to counteract the hypertensinogenic action of ouabain in rats,
- as do antibodies against ouabain, for example,
- (Digibind) and rostafuroxin (PST 2238), a selective ouabain antagonist.
It lowers BP in ouabain- and adducin-dependent hypertension in rats and is a promising new class of antihypertensive medication in humans.
REGULATION
Hypoxia
Hypoxia induces synthesis
 Endogenous Digoxin Hypoxia
Endogenous Digoxin Hypoxia
Acetylcholine (nicotinic receptor)
Endogenous ouabain-like factor (OLF) secretion is modulated by nicotinic mechanisms in rat adrenocortical cells. 2004
This study tested the hypothesis that rat adrenocortical secretion of endogenous ouabain-like factor (OLF) is regulated by nicotinic mechanisms. OLF secreted by dispersed cell suspensions of zona glomerulosa (ZG) and fasciculata/reticularis (ZFR) cells was found to co-elute with authentic ouabain by reverse phase HPLC; OLF concentrations in cell supernatants were measured by radioimmunoassay. Nicotine (10(-6) - 10(-3) M) stimulated significant OLF secretion in rat adrenocortical cells. Acetylcholine (10(-7) - 10(-4) M) and eserine (10(-7) - 10(-3) M) stimulated OLF secretion in ZG cells at lower concentrations and stimulated at higher concentrations. Acetylcholine had no effect on ZFR secretion of OLF, but eserine stimulated OLF secretion. ACTH (10(-8) M) strongly potentiated the OLF stimulatory effect of nicotine in ZG cells; however significant interactions between nicotine and ACTH or angiotensin II on OLF secretion in ZFR cells were not apparent. The ganglionic blockers hexamethonium and mecamylamine further potentiated the effect of nicotine, implicating nicotinic acetylcholine receptors (nAChRs) in regulation of OLF secretion. The alpha7-receptor antagonist methyllycaconitine (MLA) dose-dependently inhibited the effect of nicotine in the ZG cells, and in ZFR cells MLA potentiated nicotine-induced OLF secretion. These data suggest that nicotinic regulation may underlie OLF secretion by rat adrenocortical cells, and strongly suggest presence of functional nicotinic acetylcholine receptors on these cells.
Hypothalamic digoxin-mediated model for Parkinson's disease., 2003
The isoprenoid pathway produces four key metabolites important in cellular function--digoxin (endogenous membrane Na(+)-K+ ATPase inhibitor), dolichol (important in N-glycosylation of proteins), ubiquinone (free-radical scavenger), and cholesterol (component of cellular membranes). This study assessed the changes in the isoprenoid pathway and the consequences of its dysfunction in Parkinson's disease (PD). There was an elevation in plasma HMG CoA reductase activity, serum digoxin and dolichol levels, and a reduction in serum magnesium, RBC membrane Na(+)-K+ ATPase activity, and serum ubiquinone levels. Serum tryptophan, serotonin, strychnine, nicotine, and quinolinic acid were elevated, while tyrosine, morphine, dopamine, and noradrenaline were decreased. The total serum glycosaminoglycans (GAG) and glycosaminoglycan fractions (except chondroitin sulphates and hyaluronic acid), the activity of GAG degrading enzymes, carbohydrate residues of serum glycoproteins, the activity of glycohydrolase-beta galactosidase, and serum glycolipids were elevated. HDL cholesterol was reduced and free fatty acids increased. The RBC membrane glycosaminoglycans, hexose and fucose residues of glycoproteins and cholesterol were reduced, while phospholipid was increased. The activity of all serum free-radical scavenging enzymes, concentration of glutathione, alpha tocopherol, iron binding capacity, and ceruloplasmin decreased significantly in PD, while the concentration of serum lipid peroxidation products and nitric oxide increased. A dysfunctional isoprenoid pathway and related cascade are important in the pathogenesis of Parkinson's disease. A hypothalamic digoxin mediated model for ParA hypothalamic digoxin mediated model for Parkinson's disease is also postulated.

Replacement therapy
New uses for an old remedy: Digoxin as a potential treatment for steatohepatitis and other disorders, 2023
Abstract
Repurposing of the widely available and relatively cheap generic cardiac glycoside digoxin for non-cardiac indications could have a wide-ranging impact on the global burden of several diseases. Over the past several years, significant advances have been in studying digoxin pharmacology and its potential non-cardiac clinical applications, including anti-inflammatory, antineoplastic, metabolic, and antimicrobial use. Digoxin holds promise in the treatment of gastrointestinal disease, including nonalcoholic steatohepatitis and alcohol-associated steatohepatitis as well as in obesity, cancer, and treatment of viral infections, among other conditions. In this review, we provide a summary of the clinical uses of digoxin to date and discuss recent research on its emerging applications.