NADP is a cofactor used in anabolic reactions, such as the Calvin cycle and lipid and nucleic acid syntheses, which require NADPH as a reducing agent. It is used by all forms of cellular life.
NADP (Nicotinamide Adenine Dinucleotide Phosphate) exists in two forms:
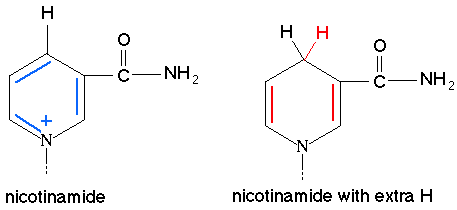
NADP+ is the oxidized form and NADPH is the reduced form.
Link 1

Reduction of NADP to NADPH
NADPH synthesis: Cytoplasm

Malate shuttle
NADPH synthesis: Mitochondria
IDH/G6PD
Glucose regulates enzymatic sources of mitochondrial NADPH in skeletal muscle cells; a novel role for glucose-6-phosphate dehydrogenase. 2010 pdf
Glucose regulates enzymatic sources of mitochondrial NADPH in skeletal muscle cells; a novel role for glucose-6-phosphate dehydrogenase. 2010
The Role of Mitochondrial NADPH-Dependent Isocitrate Dehydrogenase in Cancer Cells, 20\12
- IDH3 encodes a mitochondrial matrix NAD+-dependent octameric IDH3 (4α2β2γ subunits that acts in the Krebs cycle. IDH3 is allosterically positively regulated by Ca2+, ADP, and citrate and negatively regulated by ATP, NADH, and NADPH. The two other IDH genes, IDH1 and IDH2, encode cytosolic and mitochondrial matrix NADP+-dependent (or NADPH-dependent) IDH1 and IDH2, respectively, which are structurally and genetically unrelated to IDH3. IDH3 irreversibly decarboxylates isocitrate to yield 2OG while reducing NAD+ to NADH, whereas IDH1 and IDH2 catalyze reversible reactions, either decarboxylating isocitrate to 2OG while reducing NADP+ to NADPH or acting in the reductive carboxylation reaction to convert 2OG to isocitrate while oxidizing NADPH to NADP+. Heterozygous mutations in IDH2 at Arg172 and at the analogous residue Arg132 in IDH1 are frequently found in grade 2 and 3 gliomas, secondary glioblastomas, and acute myeloid leukemia (AML, but they occur less frequently in primary glioblastomas and other cancers. No homozygous deletions of IDH1 and IDH2 have been found, as has been observed for classic tumor suppressors. Nevertheless, mutated IDH1 and IDH2 exhibit a neomorphic enzyme activity, reducing 2OG to d-2-hydroxyglutarate while converting NADPH to NADP+. Interestingly, the d-2-hydroxyglutarate thus formed further promotes neoplasia by competitive inhibition of histone demethylation and 5-methyl-cytosine hydroxylation, leading to genome-wide alternations in the methylation of histones and DNA. It has also been reported that glioblastoma SF188 cells produce d-2-hydroxyglutarate, in spite of lacking the above-described mutations. Moreover, IDH2, like ~20% of other mitochondrial enzymes, is acetylated at lysines, which inactivates the enzymatic activity. In turn, deacetylation of IDH2 by the mitochondrial matrix deacetylase sirtuin 3 (SIRT3) activates the enzyme to produce more NADPH. In nonmalignant cells, the cytosolic IDH1 is involved in lipid metabolism and glucose sensing.
Nicotinamide Nucleotide Transhydrogenase
NNT
A significant contributor to mitochondrial NADPH is the proton-translocating transhydrogenase that catalyzes the reaction
In the presence of an electrochemical proton gradient, Δp, i.e., under physiological conditions, the reaction is strongly shifted towards NADPH formation and the rate of the reaction from left to right is enhanced 5- to 10-fold. The redox level of mitochondrial NADP is normally more than 95% reduced, about half of which is uncoupler sensitive [17]. Thus, the uncoupler-sensitive part is consistent with transhydrogenase-generated NADPH, even though contributions by other sources, e.g., the NADP-isocitrate dehydrogenase, cannot be excluded. As judged from a flux investigation of redox equivalents in E. coli under different conditions, transhydrogenase accounts for up to 45% of the NADPH flow, the remaining contributed mainly by the pentose phosphate pathway, NADP-isocitrate dehydrogenase and decarboxylating malate dehydrogenase (malic enzyme) [18]. In mitochondria, the contribution of NADPH by transhydrogenase may thus be assumed to be at least as high or higher. It should also be stressed that transhydrogenase, at the high NADPH/NADP+ redox ratio prevailing in mitochondria, has been proposed to be largely product inhibited [19], i.e., its maximal activity, approx. 30 nmol/min/mg mitochondrial protein, is only reached upon a consumption of the product NADPH at a rate exceeding that of the steady-state rate of transhydrogenase.
Mitochondrial NADPH, transhydrogenase and disease 2006
Glutamate DH
The role of the NAD-dependent glutamate dehydrogenase in restoring growth on glucose of a Saccharomyces cerevisiae phosphoglucose isomerase mutant. 1993
- Consequently, over-expression of the NAD-dependent glutamate dehydrogenase causes a substrate cycling between 2-oxoglutarate and glutamate which restores NADP from NADPH through the coupled conversion of NAD to NADH which can be oxidized in the mitochondria.
Cellular and mitochondrial effects of alcohol consumption. 2010
- There is evidence that ethanol produces alterations in the mitochondrial structure and function of several organs, including liver, and heart, both in laboratory animals and humans.
Accurate determination of the oxidative phosphorylation affinity for ADP in isolated mitochondria. 2011
- ATP Consequently, NADPH production is stoichiometrically linked to mitochondrial ATP synthesis rate, which can therefore be monitored by spectrometrically measuring the increase in the absorbance of NADPH at 340 nm.
Yeast NADH kinase
A novel NADH kinase is the mitochondrial source of NADPH in Saccharomyces cerevisiae 1993
Not yet describen in humans.
Oxidation of NADPH to NADP
- Ribonucleotide Reductase (first step of DNA synthesis)
- Fatty acid synthesis:
- >C=O => >COOH catalyzed by (3R)-3-hydroxyacyl-[acyl-carrier-protein]:NADP+ oxidoreductase, EC 1.1.1.100 (Other names: 3-oxoacyl-[acyl-carrier-protein] reductase; NADPH-specific 3-oxoacyl-[acylcarrier protein]reductase; 3-oxoacyl-[ACP]reductase; 3-ketoacyl acyl carrier protein reductase; β-ketoacyl-acyl carrier protein reductase; β-ketoacyl thioester reductase; β-ketoacyl-ACP reductase;β-ketoacyl-[acyl-carrier protein](ACP) reductase;β-ketoacyl reductase;β-ketoacyl acyl carrier protein (ACP) reductase)
* C=C => C-C catalyzed by acyl-[acyl-carrier-protein]:NAD+ oxidoreductase, EC 1.3.1.9 (other names: enoyl-[acyl-carrier-protein] reductase (NADH2; enoyl-[acyl-carrier-protein] reductase (NADH); enoyl-[acyl carrier protein] reductase; NADH-specific enoyl-ACP reductase; NADH-enoyl acyl carrier protein reductase; enoyl-ACP reductase;)
- Cholesterol biosynthesis (mevalonate pathway):
- -CO-S-CoA => -COH catalyzed by ®-mevalonate:NADP+ oxidoreductase (CoA-acylating), EC 1.1.1.34 ; HMG-CoA reductase; HMGR) 2 NADPH
- GSSG reduction to GSH
- CYP450 activity
- Proline synthesis
- Cyclooxygenase 1 and 2
- NO synthase 1, 2 and 3
Environmental factors affecting NADPH/NADP ratio
- Decrease (more oxidized status)
NADPH homeostasis in cancer: functions, mechanisms and therapeutic implications, 2020
Abstract
Nicotinamide adenine dinucleotide phosphate (NADPH) is an essential electron donor in all organisms, and provides the reducing power for anabolic reactions and redox balance. NADPH homeostasis is regulated by varied signaling pathways and several metabolic enzymes that undergo adaptive alteration in cancer cells. The metabolic reprogramming of NADPH renders cancer cells both highly dependent on this metabolic network for antioxidant capacity and more susceptible to oxidative stress. Modulating the unique NADPH homeostasis of cancer cells might be an effective strategy to eliminate these cells. In this review, we summarize the current existing literatures on NADPH homeostasis, including its biological functions, regulatory mechanisms and the corresponding therapeutic interventions in human cancers, providing insights into therapeutic implications of targeting NADPH metabolism and the associated mechanism for cancer therapy.