DESCRIPTION
Cardioactive glycosides, like digoxin, ouabain and related compounds, are drugs that inhibit Na+/K+-ATPase and have a strong inotropic effect on heart: they cause the Na+/Ca2+ exchanger to extrude Na+ in exchange with Ca2+ and therefore increase the [Ca2+]i concentration.
For this reason, some of these drugs are currently used in the treatment of congestive heart failure and cardiac arrhythmias.
CLASSIFICATION
- AA
- BB
INDICATIONS
PHARMACOKINETICS
MOLECULAR MECHANISM
PHARMACOGENOMICS
SIDE EFFECTS
TOXICITY
During February 1993-May 1995, the New York City Poison Control Center (NYCPCC) was informed about onset of illness in five previously healthy men after they ingested a substance marketed as a topical aphrodisiac; four of the men died. These cases were investigated by the New York City Department of Health, the New York City Department of Environmental Protection, and the Food and Drug Administration (FDA). Four cases were referred to the NYCPCC and one case to the New York City medical examiner's office. The decedents died from cardiac dysrhythmias, and all five patients had measurable levels of digoxin* detected in their serum. Digoxin had not been prescribed for therapeutic purposes for any of these patients, and none had medical conditions associated with endogenous digoxin-like immunoreactive substances. The purported aphrodisiac contains bufadienolides, naturally occurring cardioactive steroids that have digoxin-like effects. This report describes three of the five case reports, summarizes the investigations of the five cases, and underscores the health risks associated with inappropriate use of preparations containing digoxin-like substances.
* BB
* CC
molecular intervenction ouabain digoxin

Figure 1.
Comparison of the chemical structures of one well-known cardenolide, i.e., ouabain (A), with 2″-oxovoruscharin (UNBS1244; B) and its hemisynthetic derivative, UNBS1450 (B).
The cardenolide UNBS1450 is able to deactivate nuclear factor κB–mediated cytoprotective effects in human non–small cell lung cancer cells 2006
Cardiovascular drugs in human mechanical nociception: digoxin, amlodipine, propranolol, pindolol and atenolol. 2010
Role of endosomal Na+-K+-ATPase and cardiac steroids in the regulation of endocytosis.2007
Novel therapeutic applications of cardiac glycosides 2008

Each molecule of this family consists of three distinct structural motifs:
- a steroid nucleus
- a sugar moiety
- the sugar moiety defines the affinity for specific Na+/K+ATPase isoforms
- a lactone moiety
- the lactone moiety defines the functional class of each compound.
- Cardenolides contain a five-membered unsaturated butyrolactone ring
- Bufadienolides contain a six-membered unsaturated pyrone ring
Digoxin

Ouabain

DESCRIPTION
Digoxin is one of the cardiac glycosides, a group of drugs having in common specific effects on the myocardium. These drugs are found in a number of plants. Digoxin is extracted from the leaves of Digitalis lanata.
The glycosides are composed of two portions: a sugar and a cardenolide (hence "glycosides"). Its molecular formula is C41H64O14 and its structural formula is:

Digoxin HMD
Ouabain


A fresh facet for ouabain action 2001
INDICATIONS
Digoxin is indicated for the treatment of mild to moderate heart failure. Digoxin increases left ventricular ejection fraction and improves heart failure symptoms.
Digoxin is indicated for the control of ventricular response rate in patients with chronic atrial fibrillation. High ventricular rate leads to insufficient diastolic filling time. By slowing down the conduction in the AV node and increasing its refractory period, digoxin can reduce the ventricular rate. The arrhythmia itself is not affected, but the pumping function of the heart improves owing to improved filling.
MOLECULAR MECHANISM
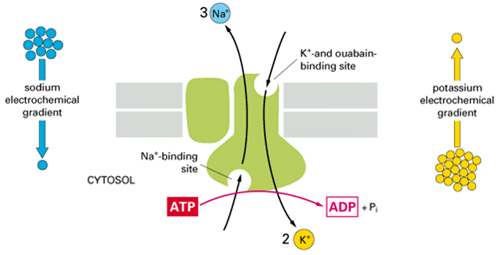
Digoxin is an inhibitor of the Na+/K+ATPase pump and it connects with its alpha subunit.
The drugs prefers to connect with the enzyme after the fosforilation of a beta aspartate,located on the surface of the alpha subunit.
During the ripolarization process Ca++ is repumped into the SR by means of a Ca++/ATPase pump and it is removed from the cell through a Na+/Ca++ exchange pump.The ability of the exchanger to expell Ca++ depends on the intracellular concentration of the Na+.The inhibition of pump Na+/K+ ATPase increases the quantity of Na+ in the cytoplasm;there is not the gradient which allows and Ca++ to be pumped outside the cell.The Ca++ in excess is taken by the SR in order to make it avaible for a new contraction.
The extracellular K+ helps the defosforilation of the enzyme during the traslocation of this cation into the cytoplasm,allowing to reduce the affinity of the enzyme for the drug.
The beneficial effects of digoxin result from direct actions on cardiac muscle, as well as indirect actions on the cardiovascular system mediated by effects on the autonomic nervous system.
The autonomic effects include
- a vagomimetic action, which is responsible for the effects of digoxin on the sinoatrial and atrioventricular nodes;
- baroreceptor sensitization, which results in increased afferent inhibitory activity and reduced activity of the sympathetic nervous system and renin-angiotensin system for any given increment in mean arterial pressure.
The pharmacologic consequences of these direct and indirect effects are:
- a positive inotropic action;
- a decrease in the degree of activation of the sympathetic nervous system and renin-angiotensin system (neurohormonal deactivating effect);
- slowing of the heart rate;
- decreased conduction velocity through the AV node (vagomimetic effect).
The effects of digoxin in heart failure are mediated by its positive inotropic and neurohormonal deactivating effects, whereas the effects of the drug in atrial arrhythmias are related to its vagomimetic actions.
In high doses, digoxin increases sympathetic outflow from the central nervous system (CNS). This increase in sympathetic activity may be an important factor in digitalis toxicity.
Ouabain induces endocytosis of plasmalemmal Na/K-ATPase in LLC-PK1 cells by a clathrin-dependent mechanism 2004

Schematic representation of the ouabain-induced endocytosis of Na/K-ATPase. Na/K-ATPase depicted show binding to ouabain (Oua, blue oval), Src (purple oval), EGFR (dark green oval), and later PIK (light green oval) and AP2 (light blue oval). Possible fates, including degradation in lysosomes, recycling to the plasmalemma, and translocation to the nucleus for signaling are illustrated.

Na+-K+–ATPase-Mediated Signal Transduction: From Protein Interaction to Cellular Function 2002
ciclo ed effetto
DOSAGE
Digoxin has a narrow therapeutic index (the margin between effectiveness and toxicity). Therapeutic serum digoxin concentrations is between 0.8 and 2.0 ng/mL.
SIDE EFFECTS
The occurrence of adverse drug reactions is common, owing to its narrow therapeutic index. Adverse effects are concentration-dependent, and are rare when plasma digoxin concentration is <0.8 μg/L.
They are also more common in patients with low potassium levels (hypokalemia), since digoxin normally competes with K+ ions for the same binding site on the Na+/K+ ATPase pump. Because some patients may be particularly susceptible to side effects with digoxin, the dosage of the drug should always be selected carefully and adjusted as the clinical condition of the patient warrants. Cardiac adverse reactions accounted for about one-half, gastrointestinal disturbances for about one-fourth, and CNS and other toxicity for about one-fourth of these adverse reactions.
High doses of digoxin may produce a variety of rhythm disturbances, such as first-degree, second-degree (Wenckebach), or third-degree heart block (including asystole); atrial tachycardia with block; unifocal or multiform ventricular premature contractions (especially bigeminy or trigeminy); ventricular tachycardia; and ventricular fibrillation. Digoxin produces PR prolongation and ST segment depression which should not by themselves be considered digoxin toxicity. Cardiac toxicity can also occur at therapeutic doses in patients who have conditions which may alter their sensitivity to digoxin. Digoxin may cause anorexia, nausea, vomiting, and diarrhea. Digoxin can produce visual disturbances (blurred or yellow vision), headache, weakness, dizziness, apathy, confusion, and mental disturbances (such as anxiety, depression, delirium, and hallucination).
Platelets
Digoxin use is associated with increased platelet and endothelial cell activation in patients with nonvalvular atrial fibrillation. 2005
Heart Rhythm. 2005 May;2(5):525-9.
Chirinos JA, Castrellon A, Zambrano JP, Jimenez JJ, Jy W, Horstman LL, Willens HJ, Castellanos A, Myerburg RJ, Ahn YS.
Comment * Heart Rhythm. 2005 May;2(5):530-1.
Abstract
OBJECTIVES: The purpose of this study was to determine whether digoxin use is associated with increased flow cytometric markers of endothelial cell and platelet activation in patients with nonvalvular atrial fibrillation (AF). BACKGROUND: Increased intracellular calcium is a key event in platelet activation, and several studies have demonstrated that digitalis activates platelets in vitro. Intracellular calcium also is a key regulator of endothelial cell function, and endogenous digitalis-like substances have been shown to affect biologic processes in endothelial cells. METHODS: We studied 30 patients with nonvalvular AF. We measured the levels of (1) platelet expression of P-selectin (CD62P), (2) platelet microparticles (PMP); and (3) endothelial microparticles (EMP) identified by anti-CD31 (EMP31) and by anti-E-selectin antibodies (EMP62E). RESULTS: Patients who were taking digoxin (n = 16; mean digoxin level = 0.93 ng/dL) did not demonstrate any significant differences in clinical or echocardiographic characteristics compared with patients not taking digoxin (n = 14). Patients taking digoxin had significantly increased levels of CD62P expression in platelets and platelet-leukocyte conjugates and markedly increased markers of endothelial activation: EMP62E and EMP31. After adjusting for potential confounders (including age, congestive heart failure, coronary artery disease, ejection fraction, antiplatelet, beta-blocker, and calcium channel blocker use), the differences persisted. CONCLUSIONS: Digoxin use in patients with AF is associated with increased levels of endothelial and platelet activation. If digitalis activates endothelial cells and platelets at pharmacologic doses, use of digitalis in conditions such as AF could predispose to thrombosis and vascular events.
TREATMENT OF ADVERSE REACTIONS
Digoxin should be temporarily discontinued until the adverse reaction resolves. Once the adverse reaction has resolved, therapy with digoxin may be reinstituted, following a careful reassessment of dose. Once the adverse reaction has resolved, therapy with digoxin may be reinstituted, following a careful reassessment of dose.
If there is a the rhythm disturbance should be given DIGIBIND [Digoxin Immune Fab (Ovine)]which is a sterile lyophilized powder of antigen binding fragments (Fab) derived from specific antidigoxin antibodies raised in sheep. DIGIBIND is a specific antidote when there is a digoxin overdosage.
It also can be given potassium canrenoate.
Every effort should be made to maintain the serum potassium concentration between 4.0 and 5.5 mmol/L.
Antiarrhythmic activity of potassium canrenoate in man.
TOXICITY
During February 1993-May 1995, the New York City Poison Control Center (NYCPCC) was informed about onset of illness in five previously healthy men after they ingested a substance marketed as a topical aphrodisiac; four of the men died. These cases were investigated by the New York City Department of Health, the New York City Department of Environmental Protection, and the Food and Drug Administration (FDA). Four cases were referred to the NYCPCC and one case to the New York City medical examiner's office. The decedents died from cardiac dysrhythmias, and all five patients had measurable levels of digoxin* detected in their serum. Digoxin had not been prescribed for therapeutic purposes for any of these patients, and none had medical conditions associated with endogenous digoxin-like immunoreactive substances. The purported aphrodisiac contains bufadienolides, naturally occurring cardioactive steroids that have digoxin-like effects. This report describes three of the five case reports, summarizes the investigations of the five cases, and underscores the health risks associated with inappropriate use of preparations containing digoxin-like substances.
* BB
* CC
RESISTANCE
DEPENDENCE AND WITHDRAW
molecular intervenction ouabain digoxin

Figure 1.
Comparison of the chemical structures of one well-known cardenolide, i.e., ouabain (A), with 2″-oxovoruscharin (UNBS1244; B) and its hemisynthetic derivative, UNBS1450 (B).
The cardenolide UNBS1450 is able to deactivate nuclear factor κB–mediated cytoprotective effects in human non–small cell lung cancer cells 2006
Cardiovascular drugs in human mechanical nociception: digoxin, amlodipine, propranolol, pindolol and atenolol. 2010
Role of endosomal Na+-K+-ATPase and cardiac steroids in the regulation of endocytosis.2007