Luca Bonomo Romeo Ayamuang
ABOUT RED MEAT
Red meat is so called for its characteristic red colour, due to the abundance of myoglobin and haemoglobin. Red meat is usually obtained by slaughtering mammals such as pigs, cows, horses, gouts and sheep.

Over the years there has been a continuous increase in demand for red meat due to an improvement in economic conditions. In fact, the meat, especially red meat, has a value of prestige in the diet and is considered a food with high nutritional value as well as a status symbol. There is a huge difference in attitudes towards meat between industrialized countries and developing ones . In fact in poor diets based on a single cereal or root, the introduction of meat and meat products, even in small quantities, can provide iron, proteins, vitamins and micronutrients which are essential for growth and development. Moreover, red meat contains a large amount of essential amino acids that are necessary to synthesize proteins and is a concentrated source of B vitamins, including vitamin B12, which is absent in plant foods. For this reason, in developing countries the tendency is to try to increase the consumption of meat, while in richer countries, where there is an excess of red meat in diet, there is a tendency to reduce the consumption. In fact, an excess of red meat in the diet is a major risk factor for several diseases such as cancer, diabetes and cardio-vascular diseases, that are major causes of mortality in the industrialized world. People are worried the meat may contain pesticides, residual of hormones and growing factors used to increase the production and they are scared by some diseases that can be transmitted to humans through meat such as Creutzfeldt-Jakob disease. Moreover meat production could lead to a higher risk of transmission of infectious diseases from animals to humans; in fact livestock densities have significantly increased, often very close to urban centres.

Colorectal Cancer Incidence and Mortality Worldwide in 2008
Meat Consumption
Dramatic changes in global meat production could increase risk of diseases
Agricultural trends, production, fertilisers, irrigation and pesticides
We are going to analyze how high red meat consumption is associated with colorectal cancer trying to give a general overview on carcinogenic molecules contained in it.
COLORECTAL CANCER
Colorectal Cancer develops in the tissues of colon (the longest part of the large intestine). Most colon cancers are adenocarcinomas (cancers that begin in cells which make and release mucus and other substances).
Cancer is a micro- evolutionary process that starts from stressful environmental conditions (in this case the conditions of the colon) in which cells accumulate damages in the genes that control reproduction and proliferation. In fact, cancer is a genetic disease of somatic cells. These genetic mutations can put cells in predisposing conditions compared to the nearby ones (there is therefore a selective pressure favouring the resistant cells and the proliferating ones). This can result in an uncontrolled growth of these cells and loss of tissue function and body homeostasis. The neoplastic progression is a process that lasts for years in which cells must change, lose or activate specific genes (oncogenes and tumour suppressor genes); most of them die but some clones, more resistant and more suitable to the stressing environment, are selected. Cancer can affect people of any age, but older people are affected more frequently, because genetic damages are accumulated during time. The most common mutations are listed below:
*the acquisition of autonomy in multiplicative process and the skill to avoid regulatory mechanisms of cell proliferation;
*the loss of density-dependent inhibition; usually cells multiply up to a defined cell density, at which they become quiescent;
*reduced ability to adhere to other cells or tissue components;
*digestion of extracellular matrix (often digested by proteases), which promotes the invasion of adjacent normal tissues;
*angiogenesis, i.e. the formation of new blood vessels to supply oxygen and nutrients to the tumor;
*reduction or loss of differentiation;
*acquisition of the capacity for unlimited replication, effect of the expression of telomerase especially in the cancer stem cells;
*reduction or loss of ability to meet programmed cell death (apoptosis) in order to avoid immune system;
*loss of so-called contact inhibition, which may lead to metastasis in other districts.
 link
link
Link interni:
flipper e nuvola-Cancer
flipper e nuvola-Cell Cycle
flipper e nuvola-Angiogenesis
flipper e nuvola-Telomere
flipper e nuvola-G1/S Checkpoint
flipper e nuvola-Nitric oxide as a bioregulator of apoptosis
flipper e nuvola-Cancer Metabolism
STATISTICAL ANALYSIS
A relationship between red meat consumption and cancer risk has been reported in a large number of epidemiological studies from many countries with diverse diets. The association between meat intake and cancer risk has been evaluated by looking both at total meat intake and finer categorizations, for example intake of red meat, such as beef, lamb and pork, and also more specifically processed meats, which includes meat preserved by salting and smoking. Processed meat differs from red meat by three major points: it often contains more fat than red meat, it contains specific additives, e.g. salt and sodium nitrate, and its long-time storage leads to cholesterol oxidation. Furthermore the process of cooking can produce more carcinogenic substances.
What seems to emerge from the bulk of works is a possible direct role of potentially carcinogenic compounds found in red meat, including nitrosamines (they require metabolic activation to be converted to a carcinogenic form), nitrosamides, heterocyclic amines and polycyclic aromatic hydrocarbons. Moreover a high red meat diet provides too much haeme, iron, salt, saturated fats and estradiol which have been accused of increasing DNA synthesis and cell proliferation, increasing insulin-like growth factors, affecting hormone metabolism and promoting free-radical damage. Other kinds of meat, such as chicken or fish, do not appear to be correlated with an increased risk of colorectal cancer.
The WCRF (World Cancer Research Fund ) recommends a maximum intake of 500 grams of red meat per week. It is also recommended to avoid processed meats completely. The data are clear: the consumption of 140 grams per day of red meat leads to an increased risk of 17% compared to no consumption at all. Doubling this amount of meat the risk increases to 34%. Studies also confirmed a very low risk for those who consume less than 70 grams daily. The risk rises exponentially if processed meat (such as bacon, ham, sausages) is considered.
"What we've found is a strong indication that many cases of bowel cancer are not inevitable, and that people are able to significantly reduce the risk by changing diet and lifestyle". Alan Jackson, President of World Cancer Research Fund.
The Associations between Food, Nutrition and Physical Activity and the Risk of Colorectal Cancer
World Cancer Research Fund International
Trends in meat consumption in the United States
Well-done Meat Intake, Heterocyclic Amine Exposure, and Cancer Risk
Meat intake and mortality: a prospective study of over half a million people
A Prospective Study of Red and Processed Meat Intake in Relation to Cancer Risk
Processed meat and colorectal cancer: a review of epidemiologic and experimental evidence
On the basis of these studies we can consider three conclusions:
# Total intake of meat is not associated with a relevant CRC risk.
# A high intake of red meat (e.g. beef, lamb and pork) is associated with significant increased risk of CRC.
# Processed meat intake, included nitrite-treated meat and meat products, is associated with CRC risk in all reports.
WHICH COMPOUNDS ARE INVOLVED?
FAT
High fat intake favours the secretion of bile acids into the duodenum; these acids, deoxycholic acid and lithocholic acid, promote colon carcinogenesis in several animal models and they are thought to be involved in human carcinogenesis. LCA (lithocholic acid) suppresses apoptosis at the base of the crypt (were stem cells are) compared with controls but does not seem to have a role in increasing cell proliferation.
A fat-rich diet may also lead to cellular damage, an effect blocked by dietary calcium. Beef tallow administration to mice causes at first (about 2 hours after administration) a damage to the intestinal epithelial cells that pass from high cylindrical cells to flatter cells with apparent signs of necrosis. This phenomenon is followed after 8 hours by a massive proliferation of cells in the intestinal crypts. Repeated mitogenic stimuli may cause an accumulation of cellular damages leading to cancer.
Membrane lipid composition and susceptibility to bile salt damage
The antiproliferative effect of dietary calcium on colonic epithelium is mediated by luminal surfactants and dependent on the type of dietary fat
The effect of lithocholic acid on proliferation and apoptosis during the early stages of colon carcinogenesis: differential effect on apoptosis in the presence of a colon carcinogen
Dietary Fatty Acids and Colorectal Cancer: A Case-Control Study
Toxicity of orally administered fat to the colonic epithelium of mice
Link interni:
flipper e nuvola-Bile Transport & Gallstones
flipper e nuvola-Ras-Raf-MEK-ERK-MAPK pathway
The vitamin D receptor (VDR) has also functions as a receptor for the secondary bile acid LCA, which is hepatotoxic and a potential enteric carcinogen. VDR is more sensitive to LCA and its metabolites than other nuclear receptors. Activation of VDR by LCA or vitamin D induces expression in vivo of CYP3A, a cytochrome P450 enzyme that detoxifies LCA in the liver and intestine. These studies offer a mechanism that may explain the proposed protective effects of vitamin D and its receptor against colon cancer.
Vitamin D Receptor As an Intestinal Bile Acid Sensor
Link interni:
flipper e nuvola-VDR
flipper e nuvola-Cytochrome P-450
NITRATE AND N-NITROSO COMPOUNDS

Nitric oxides compounds ( NOCs), like nitrosamines and nitrosamides, are alkylating agents that can react with DNA and are produced by the reaction of nitrite and nitrose oxides with secondary amines and N-alkylamides. Humans can be exposed to NOCs through consumption of certain processed meat, smoked fish and cheeses; however, a high-red meat diet can also leads to an endogenous synthesis.
There is evidence that NOCs induce the formation of DNA adduct O6-carboxymethil guanine in colonic exfoliated cells of meat-fed volunteers; this suggests that increased endogenous production of NOCs may be relevant in the etiology of colorectal cancer.
Decarboxylation of amino acids by gut bacteria produces amines and amides that can be N-nitrosated in the bowel. Haeme from meat increases NOCs production, and nitrosyl-haemoglobin is considered the major nitrosating agent in the digestive tract. Since it prevents nitrosation, ascorbic acid is often added to processed meat as an antioxidant additive.
Effect of processed and red meat on endogenous nitrosation and DNA damage
Molecular signatures of N-nitroso compounds in Caco-2 cells: implications for colon carcinogenesis
Risk of colorectal and other gastro-intestinal cancers after exposure to nitrate, nitrite and N-nitroso compounds: a follow-up study
Does increased endogenous formation of N-nitroso compounds in the human colon explain the association between red meat and colon cancer?
Haem, not protein or inorganic iron, is responsible for endogenous intestinal N-nitrosation arising from red meat
The effect of haem in red and processed meat on the endogenous formation of N-nitroso compounds in the upper gastrointestinal tract
Partial purification from hot dogs of N-nitroso compound precursors and their mutagenicity after nitrosation
Red meat enhances the colonic formation of the DNA adduct O6-carboxymethyl guanine: implications for colorectal cancer risk
Dose-dependent effect of dietary meat on endogenous colonic N-nitrosation
HETEROCYCLIC AMINES AND POLYCYCLIC AROMATIC HYDROCARBONS
HCAs and PAHs are formed during the cooking of meat. HCAs are formed by pyrolysis of creati(ni)ne with specific amino-acids. Various HCAs are formed according to the type of meat, the heating temperature and the chemical environment (water, oil, etc.). Typical HCAs in meat are 2-amino-3,8-dimethylimidazo[4,5-f] quinoxaline (MeIQx), 2-amino-3,4,9-trimethylimidazo[4,5-f]quinoxaline (DiMeIQx), and 2-amino-1-methyl-6-phenylimidazo[4,5-b]pyridine(PhIP). Nitrosation of HCAs has been proposed as a mechanism by which red meat consumption and inflammation can initiate the carcinogenesis. On the other hand, PAHs, like benzo[a]pyrene (BaP), are produced from the incomplete combustion of organic compounds.
Alkylation is the transfer of an alkyl group from one molecule to another. The alkyl group may be transferred as an alkyl carbocation, a free radical, a carbanion or a carbine. Alkylating agents are widely used in cell reactions in order to repair DNA but an excess lead to an uncontrolled and unintended reaction that damages DNA. Alkylation with only one carbon is termed methylation.

Hemin Potentiates Nitric Oxide-Mediated Nitrosation of 2-Amino-3-methylimidazo[4,5-f]quinoline (IQ) to 2-Nitrosoamino-3-methylimidazo[4,5-f]quinoline
Dietary benzo[a]pyrene intake from meat and the risk of colorectal cancer
HAEME AND IRON
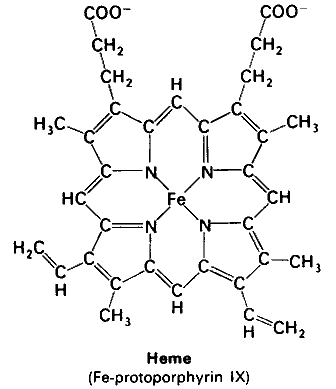
Three mechanisms may explain haeme and iron promotion of carcinogenesis:
1) Haeme catalyzes the endogenous N-nitrosation.
2) Haeme may release free iron that induces peroxydation of fat in foods and in the gut, and lipoperoxides would promote CRC.
3) Iron determines the production of ROS (Fenton reaction)

Iron is able to undergo reduction and oxidation, and these properties renders it toxic when it is present in excess. Iron can mediate the generation of reactive oxygen species (the Fenton reaction), favouring lipid peroxidation and oxidative Dna and protein damage.
In the first phase of tumorigenesis ROS and NOCs lead to mutations in key genes regulating cell proliferation, such as APC and beta catenina. In the second phase the increased cell proliferation and DNA damages cause genetic instability and an accumulation of more mutations. Iron is an important nutrient in this phase and also promotes inflammation. TNF α and IL-6, cytokines released by macrophages, increase cell survival through the activation of NFkB. During the last phase iron has been shown to activate NFkB increasing iNOS and COX-2 expression in macrophages, which produce more ROS, increase infiltration through degradation of ECM and promote angiogenesis releasing NO, PGE2, IL-1, IL-6 and IL-8.
FENTON REACTION
Fenton reaction is the reaction between hydrogen peroxide (produced during cell respiration) and an iron catalyst.
Ferrous Iron (II) is oxidized by hydrogen peroxide to ferric iron (III), a hydroxyl radical and a hydroxyl anion. Iron (III) is then reduced back to iron (II), a peroxide radical and a proton by the same hydrogen peroxide (disproportionation).
(1) Fe2+ + H2O2 → Fe3+ + OH• + OH−
(2) Fe3+ + H2O2 → Fe2+ + OOH• + H+
LIPID PEROXIDATION
Lipid peroxidation is referred to the oxidative degradation of lipids. It is the process in which free radicals "steal" electrons from the lipids in cell membranes, resulting in cell damage (mainly due to the damage of cell membranes that serve more as barriers against osmotic forces). This process often affects polyunsaturated fatty acids, because they contain multiple double bonds. As with any radical reaction, the reaction consists of three major steps: initiation, propagation, and termination.

Usually this reaction in cells starts thank to reactive oxygen species (ROS), such as OH• and HO2, which combine with a hydrogen atom to make water and a fatty acid radical. The fatty acid radical is not a very stable molecule, so it reacts readily with molecular oxygen, thereby creating a peroxyl-fatty acid radical. This is an unstable species that reacts with another free fatty acid, producing a different fatty acid radical and a lipid peroxide, or a cyclic peroxide if it had reacted with itself. This cycle continues, as the new fatty acid radical reacts in the same way. The reaction stops only when two radicals collide.
Iron: An emerging factor in colorectal carcinogenesis
The role of oxidative stress in carcinogenesis
Role of iron in carcinogenesis: cancer as a ferrotoxic disease
Signaling role of intracellular iron in NF-kappaB activation
Link interni:
flipper e nuvola-Iron Metabolism
flipper e nuvola-Heme Proteins
flipper e nuvola-NFkB
flipper e nuvola-ROS
CHOLESTEROL
Processed meat differs from common meat even for its long-time storage which may lead to cholesterol oxidation. Oxysterols are generated by enzimatic reactions mediated by cytochrome P450 family enzymes or by non-enzymatic reactions involving reactive oxygen and nitrogen species.
Oxysterols
Several studies have shown that oxysterols are involved in CRC development. A possible mechanism is the upregulation of expression of TGF-beta1 in macrophages and fibroblasts. TGF-beta1 normally promotes differentiation and apoptosis in intestinal epithelial cells. Cancer cells present a decreased expression of TGF-beta1 type I and /or type II receptors, so they are less susceptible to this factor. An increased production of TGF-beta1 thus can eliminate cancer cells more susceptible to it, favouring the selection of those cells which are insensitive and more aggressive. Moreover mutagenicity of oxysterols has been described in both nuclear and mitochondrial DNA, they can enhance ROS production through alterations in mitochondrial electron transport chain, they increase the synthesis of the pro-inflammatory cytokine IL-8 elevating its mRNA levels, 7-ketocholesterol enhances IL-1beta secretion in endothelial cells and down-regulates the expression of anti-oxidative mechanisms. To conclude some oxysterols induce COX-2 expression and are able to bind specific proteins and activate signalling cascades leading to cell migration and metastasis (for example, oxysterols-binding proteins such as ORP7 and ORP3 play a role in cell adhesion and migration through R-Ras).
Effects of oxysterols on carcinogenesis
Mechanisms of oxysterol-induced carcinogenesis
Link interni:
flipper e nuvola-TGF
flipper e nuvola-COX-1 and COX-2
CONLUSION
From what has emerged so far it seems that red meat contains many substances that can significantly increase the risk of colorectal cancer. However, much still needs to be clarified, because there are many protective factors that were not considered here. Moreover most of this substances are necessary to diet and their deficency leads to starvation and lots of other major problems. The aim of this job is not to suggest a vegetarian diet; in fact, red meat represents a significant source of proteins and other micronutrients and also plays a cultural role for some people. In conclusion we want to underline that the real risk factor is represented by processed red meat, which is associated with colorectal cancer but also with many cardiovascular diseases.